News Asia
Please note that you have to be a registered member with paid membership in order to see full articles.
RegisterSelected News

PSQ Week 2026 Begins May 18 With Quality Events Across Industries
The Philippine Society for Quality (PSQ) will hold PSQ Week 2026, its annual quality-focused celebration and learning program, from May 18-22 under the theme “57 Years of Filipino Excellence: Driving Quality Across Industries”.
TÜRKAK Revises Proficiency Testing Procedure, Issues New Subfield List
Turkish Accreditation Agency (TÜRKAK) has revised its P704 laboratory proficiency testing procedure and published a new L704-001 subfield list.
Standards Reform Proposal Aims to Cut Bureaucracy in Vietnam
The Commission for Standards, Metrology and Quality of Viet Nam (STAMEQ) has published a draft circular that would simplify administrative procedures across Vietnam’s standards, measurement, and quality infrastructure system.
BIS Releases Manak Samachar May Edition and Standards Watch 33
The Bureau of Indian Standards (BIS) has released the May edition of its Manak Samachar monthly newsletter and a new episode of its Standards Watch video series, covering recent developments in Indian standardization and workforce training.
Thailand Opens Applications for 2026 Prime Minister’s Industry Awards
Thailand’s Ministry of Industry (MIND) has opened applications for the 2026 Prime Minister’s Industry Award program,
Yad Mordechai Becomes Only Israeli Olive Oil Producer With SII Standards Mark
The Standards Institution of Israel (SII) has awarded Yad Mordechai an official Standards Mark certificate for its olive oil products, recognizing the company as the only producer currently holding the mark.
China Introduces First Provincial High-Quality Dataset Certification Program
China has introduced its first provincial-level High-Quality Dataset Inclusive Evaluation and Certification Program in Jiangsu Province, with the initiative formally launched at a conference in Wuxi on April 23.
Spring 2026 ‘ANSI in China’ Newsletter Highlights Standards and Policy Developments
The American National Standards Institute (ANSI) has published the Spring 2026 edition of its ANSI in China Newsletter in English and Chinese.
Korea Accreditation Board Updates ISO 19443 Nuclear Supply Chain Certification Rules
The Korea Accreditation Board (KAB) has revised its certification scheme requirements for ISO 19443, introducing updated rules for certification bodies auditing quality management systems (QMS) in the nuclear supply chain.
Vietnam Sets Transition Dates for ISO/IEC 17020:2026
The National Accreditation Bureau of Vietnam (BoA) has announced the transition schedule for ISO/IEC 17020:2026, the new edition of the conformity assessment standard for inspection bodies replacing ISO/IEC 17020:2012.
ISO 59020 Revision Starts With Indian Experts in Prominent Roles
Work has officially started within ISO/TC 323 Working Group 3 on revising ISO 59020, the international standard for measuring and assessing circularity performance.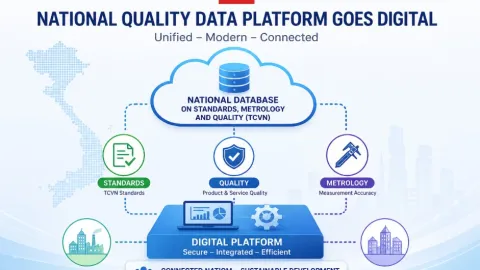
Vietnam Establishes National Database for Standards, Metrology and Quality
The Commission for Standards, Metrology and Quality of Viet Nam (STAMEQ) has defined a unified national database for standards, metrology and quality underDecree 22/2026/ND-CP,Global News
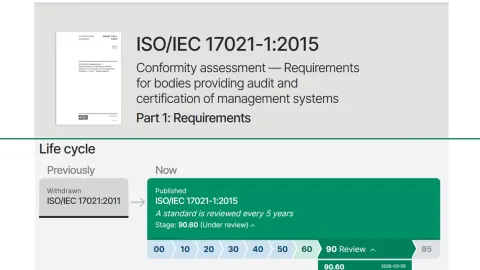
ISO/IEC 17021-1 Confirmation Keeps Certification Rules Stable
The International Organization for Standardization’s (ISO) Committee on Conformity Assessment (ISO/CASCO) has confirmed continuation of the current edition of ISO/IEC 17021-1,
Revised ISO 14024 Aims to Prevent Greenwashing in Ecolabel Programs
The International Organization for Standardization (ISO) has published the third edition of ISO 14024, revising requirements for ecolabel certification programs to improve verification of environmental claims and help prevent greenwashing,
Experts Sought for ISO TS 20008 Food Waste Measurement and Reporting Project
The International Organization for Standardization (ISO) has started development of ISO/AWI TS 20008,
Joint ISO and IEC Team Releases Information Model for SMART Content
The International Organization for Standardization (ISO) and the International Electrotechnical Commission (IEC) have released the first version of the IEC & ISO Information Model for SMART content,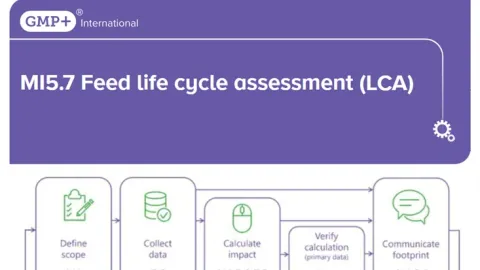
GMP+ International Launches Feed Life Cycle Assessment Standard
The GMP+ International has launched the MI5.7 Feed Life Cycle Assessment LCA standard, introducing a harmonized method for calculating and communicating the environmental impact of feed products across the supply chain.
ISO 22367:2026 Revises Risk Management Guidance for Medical Laboratories
The International Organization for Standardization (ISO) has published ISO 22367:2026, a revised standard that provides medical laboratories with a structured approach to identifying, assessing, and controlling risks across laboratory activities and testing processes.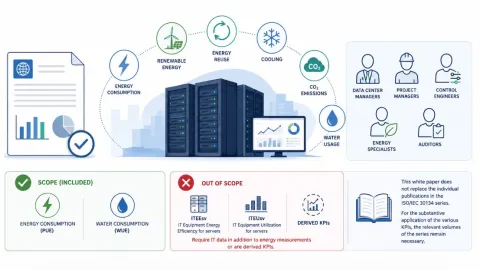
ISO 30134 Series KPIs Highlighted in New Data Center Energy and Water Whitepaper
A new whitepaper published by the International Organization for Standardization (ISO) explains how key performance indicators (KPIs) from the ISO/IEC 30134 series can be used to measure and report energy and water use in data centers.
CSQ Invites Comments on Cannabis EU GMP Certification Addendum
Cannabis Safety & Quality (CSQ) has opened a public comment period for a new EU GMP certification addendum,
FSSC 22000 Version 7 Updates Prerequisite Programs And Aligns With GFSI 2024
The Foundation FSSC has released Version 7 of the FSSC 22000 food safety management system certification scheme, introducing updated prerequisite programs (PRPs) and aligning the scheme with the 2024 Global Food Safety Initiative requirements.
ISO Develops Carbon Credit Data Model Standard, Calls for Input
A new data model for carbon credit markets is being developed by ISO’s sustainable finance and financial services committees, with early input still open to stakeholders.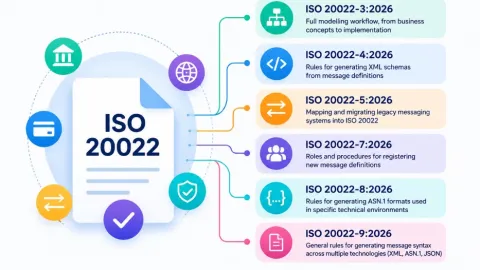
ISO 20022 Series Update Sets New Rules as Payment Systems Prepare for Implementation
The International Organization for Standardization (ISO) has published updated and new parts in the ISO 20022 series, as major payment systems advance their implementation of the standard on closely aligned timelines.

