News China
Please note that you have to be a registered member with paid membership in order to see full articles.
RegisterSelected News

China Introduces First Provincial High-Quality Dataset Certification Program
China has introduced its first provincial-level High-Quality Dataset Inclusive Evaluation and Certification Program in Jiangsu Province, with the initiative formally launched at a conference in Wuxi on April 23.
Spring 2026 ‘ANSI in China’ Newsletter Highlights Standards and Policy Developments
The American National Standards Institute (ANSI) has published the Spring 2026 edition of its ANSI in China Newsletter in English and Chinese.
China Introduces First Commercial Space Standards System Covering Full Industry Chain
The China National Space Administration (CNSA) and the State Administration for Market Regulation (SAMR) have released the Commercial Space Standard System (Version 1.0), China’s first structured framework to guide the country’s growing private space sector.
China Seeks Experts For International Standards On Battery Recycling And Humanoid Robot Data
The Standards Innovation Department of the State Administration for Market Regulation (SAMR) is inviting experts to join national groups supporting two international standards, one on handling end-of-life power batteries and another on datasets used to train humanoid robots.
China Targets AI, Data And Advanced Manufacturing in New National Standardization Pilot Program
The Standardization Administration of China (SAC) has opened applications for its 2026 national standardization pilot program,
China Updates Laboratory Accreditation Rules for Medical Device Testing
China National Accreditation Service for Conformity Assessment (CNAS) has issued a revised version of its guidance for applying laboratory accreditation criteria in medical device testing, with CNAS-CL01-A004:2026 set to take effect on May 1, 2026.
China Issues New Guide For Public Institution Carbon Accounting
A new industry standard for carbon accounting in public institutions took effect on April 1, marking a step toward more consistent measurement and management of emissions across China’s public sector.
China Sets Up Seven Standardization Bodies Including Humanoid Robots Group
The Standardization Administration of China (SAC) has announced the creation of seven new national technical committees and subcommittees, expanding standardization work across sectors from agriculture to advanced technologies.
China Tightens GMP Rules For Medical Device Manufacturers Effective November 2026
The National Medical Products Administration (NMPA) has revised China’s Good Manufacturing Practice (GMP) requirements for medical devices, introducing stricter quality management system rules that will take effect on November 1, 2026 and replace the current 2014 version.
TIC Industry Conference in Beijing Focuses on Trust, Restructuring, and Global Standards Role
The China Quality Certification Center (CQC) hosted the 2026 annual conference on testing, inspection, and certification (TIC) in Beijing on March 26, focusing on strengthening trust, supporting trade, and guiding industry transformation.
China Sets Up Three Regional AI Standardization Groups With Distinct Roles
The Standardization Administration of China (SAC) has established three regional working groups for artificial intelligence, each with a defined focus ranging from international standards work to industrial application.
China Organizes National Push For Codex Agricultural Proposals
China has launched a national call for proposals to support its participation in Codex Alimentarius Commission (CAC) work in the agricultural sector,Global News

ISO 9001 Revision Moves Into Formal FDIS Ballot Phase
The International Organization for Standardization (ISO) has advanced the revision of ISO 9001 to the next step of the Final Draft International Standard (FDIS) approval process, with the project now listed at stage 50.20 following the earlier 50.00 status.
FSC Opens Consultation on Chain of Custody Revisions as Two Core Standards Reach Final Phase
The Forest Stewardship Council (FSC) has opened consultation on revisions to five Chain of Custody (CoC) documents, with two core standards now entering the final consultation phase.
CanadaGAP Seeks GFSI Recognition for Updated Food Safety Programs
The Global Food Safety Initiative (GFSI) has opened a public stakeholder consultation as CanadaGAP seeks recognition for updated food safety certification programs covering fruit and vegetable operations.
Efforts to Broaden Application of ISO 21378 Underway
Following last year’s confirmation of ISO 21378:2019, a standard for structured audit data collection and exchange, work within ISO/TC 295/WG 1 is now focused on broadening how the standard is applied across organizations.
ISO 14019-3 Working Draft Reviewed Ahead of October Stage Decision
The development of ISO 14019-3, a proposed international standard on sustainability information validation processes,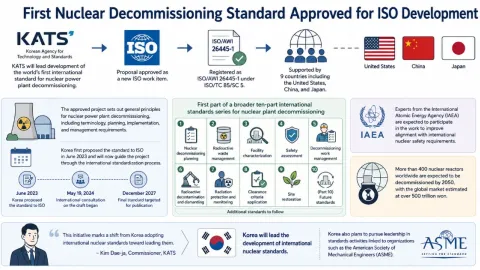
First Nuclear Decommissioning Standard Approved for ISO Development
The Korean Agency for Technology and Standards (KATS) will lead development of the world’s first international standard for nuclear power plant decommissioning after its proposal was approved as a new ISO work item.
Public Consultation Opens for Revised ISO 19650-3 BIM Standard
A public consultation has opened for the revised draft of ISO 19650-3, the part of the ISO 19650 series focused on information management during the operational phase of built assets.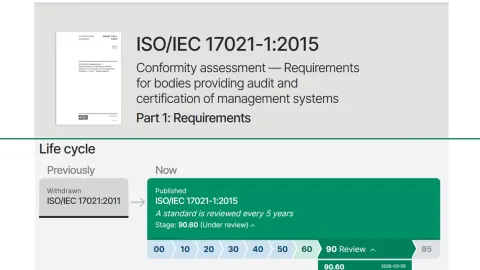
ISO/IEC 17021-1 Confirmation Keeps Certification Rules Stable
The International Organization for Standardization’s (ISO) Committee on Conformity Assessment (ISO/CASCO) has confirmed continuation of the current edition of ISO/IEC 17021-1,
Revised ISO 14024 Aims to Prevent Greenwashing in Ecolabel Programs
The International Organization for Standardization (ISO) has published the third edition of ISO 14024, revising requirements for ecolabel certification programs to improve verification of environmental claims and help prevent greenwashing,
Experts Sought for ISO TS 20008 Food Waste Measurement and Reporting Project
The International Organization for Standardization (ISO) has started development of ISO/AWI TS 20008,
Joint ISO and IEC Team Releases Information Model for SMART Content
The International Organization for Standardization (ISO) and the International Electrotechnical Commission (IEC) have released the first version of the IEC & ISO Information Model for SMART content,