News United States of America
Please note that you have to be a registered member with paid membership in order to see full articles.
RegisterSelected News

NIST Expands AI Consortium, Invites New Organizations
The National Institute of Standards and Technology (NIST) has renamed its AI Safety Institute Consortium as the NIST AI Consortium,
Joint Commission Launches Healthcare Cyber Resilience Program With Future Certification Pathway
The Joint Commission, a U.S. healthcare accreditation body, and the American Hospital Association (AHA) have launched a new Cyber Resilience Readiness (CRR) program for healthcare organizations.
Current Standards System at Risk After IBR Court Decisions, ANSI Brief Warns
The American National Standards Institute (ANSI) is warning standards developing organizations (SDOs) that recent US court decisions involving standards incorporated by reference (IBR) could threaten the long-standing system used to fund, maintain, and update voluntary standards.
NIST Pilot Program Aims to Accelerate Aerospace Manufacturing and Critical Minerals Supply Chains
The National Institute of Standards and Technology (NIST) has announced a new pilot program aimed at accelerating advanced manufacturing technologies in aerospace component production and the domestic critical minerals supply chain.
ANSI Honors Walter Zoller With Ronald H. Brown Standards Leadership Award
The American National Standards Institute (ANSI) has selected Walter Zoller, senior manager of technology standards and regulations at Rockwell Automation, as the 2026 recipient of the Ronald H. Brown Standards Leadership Award.
Students Invited to Explore a World Without Standards in ANSI Paper Competition
The American National Standards Institute (ANSI) is reminding high school and college students across the United States to enter its 2025/2026 Student Paper Competition by June 5, 2026.
CPSC Opens Consultation on Counterfeit Safety Certification Marks
The U.S. Consumer Product Safety Commission (CPSC) is seeking public comments on counterfeit certification marks used on consumer products,
ANSI and America Makes Report Tracks Progress on 35 Additive Manufacturing Standards Gaps
The American National Standards Institute (ANSI) and manufacturing innovation institute America Makes have released a new report on additive manufacturing standards gaps that shows progress on 35 of the 141 gaps identified in their 2023 roadmap.
Spring 2026 ‘ANSI in China’ Newsletter Highlights Standards and Policy Developments
The American National Standards Institute (ANSI) has published the Spring 2026 edition of its ANSI in China Newsletter in English and Chinese.
ANSI Starts National Effort To Align Critical Minerals Standards Across The Supply Chain
The American National Standards Institute (ANSI) has launched a multi-phase initiative to coordinate standards for critical minerals, aiming to strengthen U.S. supply chains and reduce reliance on foreign sources.
Quantum Standards Lag Behind Technology Growth, ASCET Scan Finds
The Advancing Standardization for Critical and Emerging Technologies (ASCET) Center of Excellence (CoE) has found in a new environmental scan that quantum standardization is not keeping pace with rapid technological progress,
ANAB Launches QAEP to Evaluate Credibility and Effectiveness of Quality Assurance Schemes
The ANSI National Accreditation Board (ANAB) has launched the Quality Assurance Evaluation Program (QAEP),Global News

ISO 9000 and ISO 19011 Revised: New Guidance for Quality and Auditing
Two cornerstone standards for quality management and auditing have been revised, with the International Organization for Standardization (ISO) publishing new editions of ISO 9000 and ISO 19011.
ISO 9002 Revision Reaches Public Comment Stage
The International Organization for Standardization (ISO) has advanced the revision of ISO 9002 to the Draft International Standard (DIS) stage, opening the project for wider review and feedback.
Global ACI Sets 36-Month Transition for ISO/IEC 17024:2026
The Global Accreditation Cooperation Incorporated (Global ACI) has established a 36-month transition period for ISO/IEC 17024:2026, the newly revised international standard for bodies that certify individuals.
PAS 96:2026 Updates Food Defence Guidance for Emerging Threats
The British Standards Institution (BSI) has published PAS 96:2026, a fully revised edition of its food defence guide.
BRCGS Updates Meat Supply Chain Assurance Module
The British Retail Consortium Global Standards (BRCGS) has updated its Additional Module 11: Meat Supply Chain Assurance.
ISO 29997 Offers First International Framework for Quality Internships
ISO 29997:2025 provides the first international framework designed to help organizations deliver higher-quality internships through clearer planning, management and evaluation practices.
Experts Behind ISO 14001:2026 Explain New Focus on Climate, Biodiversity and Results
The International Organization for Standardization (ISO) has outlined the most important changes in ISO 14001:2026,
New ISO/IEC 15408 and 18045 Editions Update Common Criteria Framework
ISO/IEC 15408-1:2026 and ISO/IEC 18045:2026 have been published, updating the Common Criteria framework, an international system used to evaluate the security of IT products.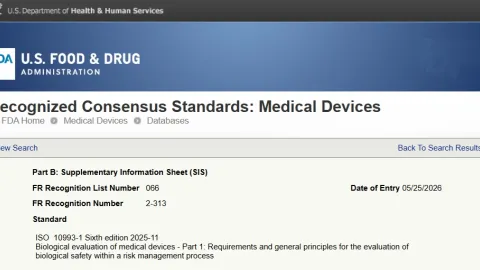
FDA Partially Recognizes ISO 10993-1:2025 for Medical Devices
The U.S. Food and Drug Administration (FDA) has partially recognized ISO 10993-1:2025, the latest edition of the international standard used to assess the biological safety of medical devices.
Licensing Instead of Sales: ISO Changes How Standards Are Used
The International Organization for Standardization (ISO) is preparing to replace its traditional standards sales approach with a licensing model that would give users defined rights to use standards content instead of broad unrestricted access.
BRCGS Updates Food Safety, Packaging, Storage, and Broker Rules to Meet New GFSI Requirements
BRCGS has updated certification requirements for its Food Safety, Packaging Materials, Storage and Distribution, and Agents and Brokers standards,

