News Medicine / Pharmaceutical
Please note that you have to be a registered member with paid membership in order to see full articles.
RegisterSelected News

EA Updates Guidance for Assessing Point-of-Care Testing Under EN ISO 15189
European co-operation for Accreditation (EA) has published a revised version of EA-4/20, updating how laboratories and accreditation bodies assess point-of-care testing (POCT) under EN ISO 15189:2022.
Cofrac Updates Map Of Accredited Medical Biology Laboratories In France
The French Accreditation Committee (Cofrac) has released an updated map showing the distribution of accredited medical biology laboratories (LBM) across France as of April 1, 2026.
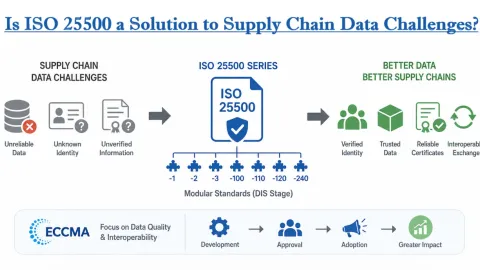
China Updates Laboratory Accreditation Rules for Medical Device Testing
China National Accreditation Service for Conformity Assessment (CNAS) has issued a revised version of its guidance for applying laboratory accreditation criteria in medical device testing, with CNAS-CL01-A004:2026 set to take effect on May 1, 2026.
China Tightens GMP Rules For Medical Device Manufacturers Effective November 2026
The National Medical Products Administration (NMPA) has revised China’s Good Manufacturing Practice (GMP) requirements for medical devices, introducing stricter quality management system rules that will take effect on November 1, 2026 and replace the current 2014 version.
India Invites MSMEs to Join Healthcare Standards Committees
The Bureau of Indian Standards (BIS) is inviting micro, small, and medium enterprises (MSMEs) to join technical committees in its Medical Equipment and Hospital Planning Department (MHD), opening the standardization process to a wider range of industry participants.
Dutch Healthcare Information Security Standard Updated to Align With ISO 27799
The Royal Netherlands Standardization Institute (NEN) has published an amendment to NEN 7510-2 to align it with ISO 27799,
China Clarifies Registration Rules for Special Medical Foods Under New Standards
The State Administration for Market Regulation (SAMR) has issued a Q&A to explain how foods for special medical purposes (FSMP) should be registered under China’s updated national standards, helping companies understand how to apply the new rules in practice.
AKH Wien Becomes First Hospital In Austria Certified For Diversity Management
The Vienna General Hospital (AKH Wien) has become the first hospital in Austria to receive certification for its diversity management system,
FDA Schedules Town Hall On Risk Based Inspections Under New QMSR Framework
The U.S. Food and Drug Administration (FDA) has announced a Town Hall on April 1, 2026, to explain how inspections of medical device manufacturers will change under its new Quality Management System Regulation (QMSR), with a focus on a risk-based approach.
Mountain Healthcare First In UK To Achieve ISO 15189 Accreditation For SARC Services
Mountain Healthcare has become the first provider in the UK to achieve accreditation from the United Kingdom Accreditation Service (UKAS) to ISO 15189:2022 for its Sexual Assault Referral Centres (SARCs),
FDA Shifts Medical Device Inspections To Lifecycle-Based QMS Oversight
The U.S. Food and Drug Administration (FDA) has updated how it inspects medical device manufacturers, introducing a lifecycle‑based approach to quality management system oversight under its new Quality Management System Regulation.
Free Risk Tool Launched for Healthcare Information Security Standard NEN 7510
The Netherlands Standardization Institute (NEN) has launched a free risk analysis tool to help healthcare organizations and other entities that process personal health data carry out the information security risk assessment required under NEN 7510.Global News

FSSC 22000 Version 7 Updates Prerequisite Programs And Aligns With GFSI 2024
The Foundation FSSC has released Version 7 of the FSSC 22000 food safety management system certification scheme, introducing updated prerequisite programs (PRPs) and aligning the scheme with the 2024 Global Food Safety Initiative requirements.
ISO Develops Carbon Credit Data Model Standard, Calls for Input
A new data model for carbon credit markets is being developed by ISO’s sustainable finance and financial services committees, with early input still open to stakeholders.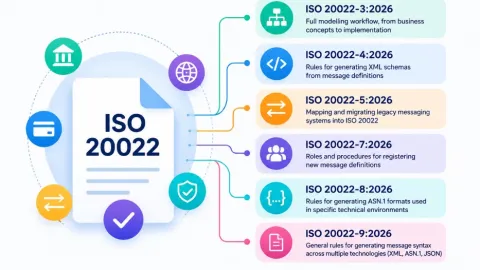
ISO 20022 Series Update Sets New Rules as Payment Systems Prepare for Implementation
The International Organization for Standardization (ISO) has published updated and new parts in the ISO 20022 series, as major payment systems advance their implementation of the standard on closely aligned timelines.
Consultation Underway On Global Standard For AI Testing In Medical Devices
An international voting and consultation phase is underway for a new standard that sets requirements for testing artificial intelligence (AI) and machine learning (ML) in medical devices.
MSC Opens Review Of Chain Of Custody Standard For Public Consultation
The Marine Stewardship Council (MSC) has launched a public consultation on proposed revisions to its Chain of Custody Standard, inviting stakeholders across the seafood supply chain to provide feedback until May 30, 2026.
Call For Food Sector Stakeholders To Influence ISO 22000 Revision Through National Committees
Food sector stakeholders who apply ISO 22000 in practice have the opportunity to influence its revision by bringing real-world experience into the process.
AutoCarbon Certification Scheme Due in 2027 As Pilot Phase Begins
The International Automotive Oversight Bureau (IAOB) has announced AutoCarbon,
ISO 14064-5 Introduces Guidance For Remote Verification Of Greenhouse Gas Data
The International Organization for Standardization (ISO) has introduced ISO 14064-5, which provides guidance on how to carry out verification and validation of greenhouse gas (GHG) data using remote techniques in digital environments.
ISO 9002 Revision Moves To Draft International Standard Stage
The revision of ISO 9002 has entered the Draft International Standard (DIS) stage, with ISO/TC 176/SC 2, the ISO subcommittee responsible for quality systems, advancing the project into formal consultation.
ISO 37304 Compliance Management Certification Standard Advances to Final Draft Stage
The International Organization for Standardization (ISO) has advanced ISO 37304 to the final draft international standard (FDIS) stage.
One Vote Tips Toward New Edition Of ISO/IEC 17065 Over Amendment
One vote has tipped the decision toward publishing a new edition of ISO/IEC 17065 instead of issuing a separate amendment, following a closely split ballot within the International Organization for Standardization (ISO).