News Medicine / Pharmaceutical
Please note that you have to be a registered member with paid membership in order to see full articles.
RegisterSelected News
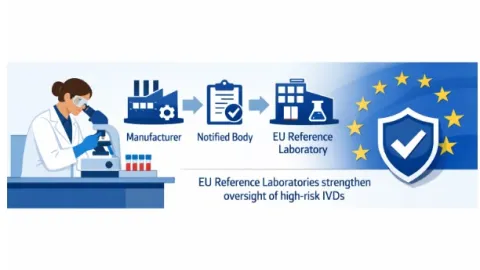
EU Expands Laboratory Oversight for Highest Risk In Vitro Diagnostic Devices
The European Union (EU) has expanded oversight of Class D in vitro diagnostic medical devices under Regulation (EU) 2017/746, known as the In Vitro Diagnostic Regulation (IVDR).
BIS Seeks Public Feedback on Draft Standards for Medical Devices and Healthcare
The Bureau of Indian Standards (BIS), through its Medical Equipment and Hospital Planning Department,
NATA Seeks Experts in Software as Medical Devices and AI for Human Pathology Advisory Committee
The National Association of Testing Authorities, Australia (NATA) is inviting expressions of interest to join its Human Pathology Accreditation Advisory Committee,
Denmark Aligns Accreditation Rules for Referral Laboratories and Specialists With ISO 15189
Denmark has revised its national accreditation rules for the use of referral laboratories and external specialists to align with the Danish version of ISO 15189:2022 for medical laboratories.
FDA Aligns U.S. Device Rules With ISO 13485, Replaces QSR With QMSR
The U.S. Food and Drug Administration (FDA) has replaced its long-standing Quality System Regulation (QSR) with the new Quality Management System Regulation (QMSR), changing the legal framework for quality management in the U.S. medical device sector.
NEN Seeks Experts for Clinical Research Standards Committee
The Netherlands Standardization Institute (NEN) is inviting experts to join its Clinical Research standards committee to help shape international standards for clinical investigations of medical devices.
Clearlab Earns MDSAP, ISO 13485 and EU MDR Certifications for Contact Lenses
Clearlab, a manufacturer of vision correction products and lens care solutions, has obtained combined certification under the Medical Device Single Audit Program (MDSAP), ISO 13485, and the European Union Medical Device Regulation (EU MDR).
EU Medical Device Database EUDAMED Opens First Mandatory Modules
The European Commission has published the first four functional modules of EUDAMED, the EU database for medical devices and in vitro diagnostic devices.
Italy Launches Accreditation for ISO 7101 Healthcare Quality Systems
Italy has opened accreditation for certification bodies that audit healthcare organizations against UNI ISO 7101:2024, the standard for quality management in health services.
IEI Secures TFDA QMS Approval and ISO 13485 Certification
IEI Integration Corp, a Taiwan-based provider of medical and industrial computing systems,
NEN Sets Initial Scope For New Guideline On Sustainable Medical Devices
The Netherlands Standardization Institute (NEN) has set the initial scope for developing a new Dutch Practice Guideline (NPR) on sustainability in medical devices.
Mexico Recognizes MDSAP as Equivalent to National Medical Device GMP
Mexico has issued a new interpretation rule for the NOM-241-SSA1-2025 standard on good manufacturing practices (GMP) for medical devices, confirming thatGlobal News

ISO/TS 19384 Sets First Standard for Digital Service Excellence
The International Organization for Standardization (ISO) has released ISO/TS 19384:2026,
FSSC Issues Five Sector-Specific Guides for Development Program Implementation
The Foundation for Food Safety Certification (FSSC) has published five sector-specific guidance documents to support organizations using the FSSC Development Program,
ISO 14001 Fourth Edition Officially Released
The International Organization for Standardization (ISO) has published ISO 14001:2026, marking the release of the fourth edition of the world’s most widely used environmental management systems standard.
Global ACI Introduced: IAF and ILAC Successor Presents New Identity
A new international accreditation body, Global Accreditation Cooperation Incorporated, has introduced a new identity as Global ACI, including a logo and communication channels after starting operations on January 1, 2026.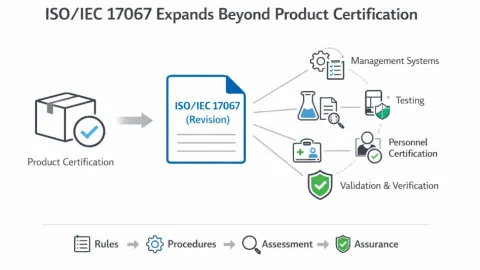
ISO 17067 Revision Expands Conformity Assessment Schemes
The International Organization for Standardization (ISO) Committee on Conformity Assessment (CASCO) is revising ISO/IEC 17067 to expand its scope from product certification to a broader framework for conformity assessment schemes.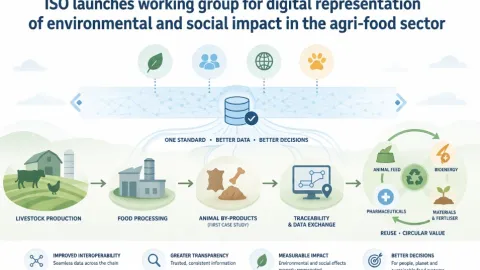
ISO Establishes Working Group on Environmental and Social Impact Data in Agrifood Systems
The International Organization for Standardization (ISO) has set up a new working group under ISO/TC 347 to develop a standardized way to record and share data on environmental and social impacts across agrifood supply chains.
European AI Standard for Computer Vision prEN 18281 Enters Public Enquiry
A European draft standard, prEN 18281, for evaluating AI systems that analyze images and video has entered public enquiry, setting out how such systems should be assessed as part of the broader framework supporting the EU AI Act.
Work Begins on IEC 60601-1 Medical Electrical Equipment Safety Standard Update
The International Electrotechnical Commission (IEC) has begun work on updating IEC 60601-1, the core standard for the safety and essential performance of medical electrical equipment.
ISSB Proposes Updates to Three SASB Standards, Refining Sustainability Reporting in Agriculture, Food and Power
The International Sustainability Standards Board (ISSB) has issued a consultation on proposed amendments to three Sustainability Accounting Standards Board (SASB) Standards to support alignment with IFRS S1 and IFRS S2,
FSC Proposes New Approach To Intact Forest Landscapes Protection
The Forest Stewardship Council (FSC) has launched two public consultations on draft Advice Notes to strengthen how Intact Forest Landscapes (IFLs) are managed and protected.
VDA Releases Automotive SPICE Yellow Volume 3rd Revised Edition for Public Review
The German Association of the Automotive Industry (VDA) Quality Management Center (QMC) has released the Automotive SPICE Yellow Volume 3rd revised edition, opening a public feedback period until May 12, 2026.

