News Medicine / Pharmaceutical
Please note that you have to be a registered member with paid membership in order to see full articles.
RegisterSelected News

Free Risk Tool Launched for Healthcare Information Security Standard NEN 7510
The Netherlands Standardization Institute (NEN) has launched a free risk analysis tool to help healthcare organizations and other entities that process personal health data carry out the information security risk assessment required under NEN 7510.
Hoan My International Hospital Laboratory Earns ISO 15189 Accreditation
The medical laboratory of Hoan My International Hospital has received ISO 15189:2022 accreditation from the National Accreditation Bureau (BoA),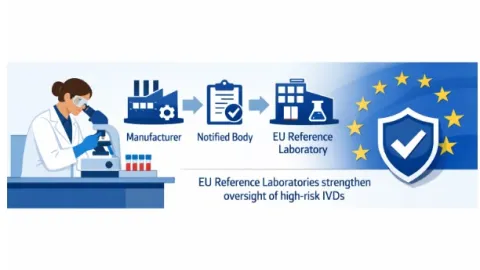
EU Expands Laboratory Oversight for Highest Risk In Vitro Diagnostic Devices
The European Union (EU) has expanded oversight of Class D in vitro diagnostic medical devices under Regulation (EU) 2017/746, known as the In Vitro Diagnostic Regulation (IVDR).
BIS Seeks Public Feedback on Draft Standards for Medical Devices and Healthcare
The Bureau of Indian Standards (BIS), through its Medical Equipment and Hospital Planning Department,
NATA Seeks Experts in Software as Medical Devices and AI for Human Pathology Advisory Committee
The National Association of Testing Authorities, Australia (NATA) is inviting expressions of interest to join its Human Pathology Accreditation Advisory Committee,
Denmark Aligns Accreditation Rules for Referral Laboratories and Specialists With ISO 15189
Denmark has revised its national accreditation rules for the use of referral laboratories and external specialists to align with the Danish version of ISO 15189:2022 for medical laboratories.
FDA Aligns U.S. Device Rules With ISO 13485, Replaces QSR With QMSR
The U.S. Food and Drug Administration (FDA) has replaced its long-standing Quality System Regulation (QSR) with the new Quality Management System Regulation (QMSR), changing the legal framework for quality management in the U.S. medical device sector.
NEN Seeks Experts for Clinical Research Standards Committee
The Netherlands Standardization Institute (NEN) is inviting experts to join its Clinical Research standards committee to help shape international standards for clinical investigations of medical devices.
Clearlab Earns MDSAP, ISO 13485 and EU MDR Certifications for Contact Lenses
Clearlab, a manufacturer of vision correction products and lens care solutions, has obtained combined certification under the Medical Device Single Audit Program (MDSAP), ISO 13485, and the European Union Medical Device Regulation (EU MDR).
EU Medical Device Database EUDAMED Opens First Mandatory Modules
The European Commission has published the first four functional modules of EUDAMED, the EU database for medical devices and in vitro diagnostic devices.
Italy Launches Accreditation for ISO 7101 Healthcare Quality Systems
Italy has opened accreditation for certification bodies that audit healthcare organizations against UNI ISO 7101:2024, the standard for quality management in health services.
IEI Secures TFDA QMS Approval and ISO 13485 Certification
IEI Integration Corp, a Taiwan-based provider of medical and industrial computing systems,Global News

Revised ISO 14024 Aims to Prevent Greenwashing in Ecolabel Programs
The International Organization for Standardization (ISO) has published the third edition of ISO 14024, revising requirements for ecolabel certification programs to improve verification of environmental claims and help prevent greenwashing,
Experts Sought for ISO TS 20008 Food Waste Measurement and Reporting Project
The International Organization for Standardization (ISO) has started development of ISO/AWI TS 20008,
Joint ISO and IEC Team Releases Information Model for SMART Content
The International Organization for Standardization (ISO) and the International Electrotechnical Commission (IEC) have released the first version of the IEC & ISO Information Model for SMART content,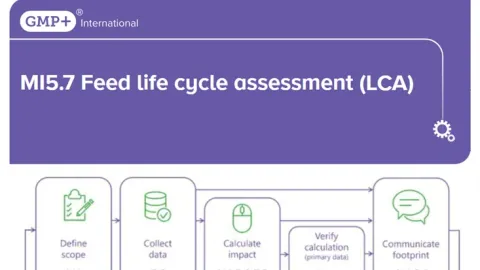
GMP+ International Launches Feed Life Cycle Assessment Standard
The GMP+ International has launched the MI5.7 Feed Life Cycle Assessment LCA standard, introducing a harmonized method for calculating and communicating the environmental impact of feed products across the supply chain.
ISO 22367:2026 Revises Risk Management Guidance for Medical Laboratories
The International Organization for Standardization (ISO) has published ISO 22367:2026, a revised standard that provides medical laboratories with a structured approach to identifying, assessing, and controlling risks across laboratory activities and testing processes.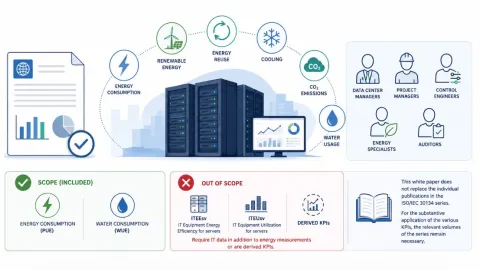
ISO 30134 Series KPIs Highlighted in New Data Center Energy and Water Whitepaper
A new whitepaper published by the International Organization for Standardization (ISO) explains how key performance indicators (KPIs) from the ISO/IEC 30134 series can be used to measure and report energy and water use in data centers.
CSQ Invites Comments on Cannabis EU GMP Certification Addendum
Cannabis Safety & Quality (CSQ) has opened a public comment period for a new EU GMP certification addendum,
FSSC 22000 Version 7 Updates Prerequisite Programs And Aligns With GFSI 2024
The Foundation FSSC has released Version 7 of the FSSC 22000 food safety management system certification scheme, introducing updated prerequisite programs (PRPs) and aligning the scheme with the 2024 Global Food Safety Initiative requirements.
ISO Develops Carbon Credit Data Model Standard, Calls for Input
A new data model for carbon credit markets is being developed by ISO’s sustainable finance and financial services committees, with early input still open to stakeholders.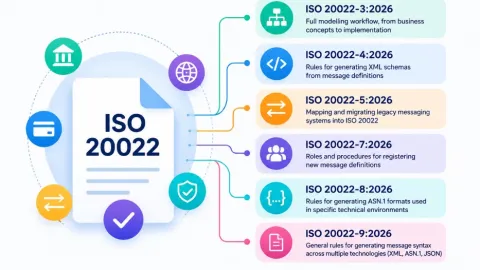
ISO 20022 Series Update Sets New Rules as Payment Systems Prepare for Implementation
The International Organization for Standardization (ISO) has published updated and new parts in the ISO 20022 series, as major payment systems advance their implementation of the standard on closely aligned timelines.
Consultation Underway On Global Standard For AI Testing In Medical Devices
An international voting and consultation phase is underway for a new standard that sets requirements for testing artificial intelligence (AI) and machine learning (ML) in medical devices.

