News Netherlands
Please note that you have to be a registered member with paid membership in order to see full articles.
RegisterSelected News

Dutch Initiative Prepares New Committee on Medical Device Environmental Impact
The Royal Netherlands Standardization Institute (NEN) is preparing a new committee focused on methods for assessing the environmental impact of medical devices, with the aim of helping shape Dutch input into future European and international standardization work.
NEN Seeks Members for Flexible, Demountable Infrastructure Standards
The Netherlands Standardization Institute (NEN) is seeking members for new working groups that will further develop three Dutch Technical Agreements (NTAs) related to industrial, flexible, and demountable (IFD) infrastructure construction methods.
WMDA Earns Accreditation for Global Stem Cell Donor Certification
The World Marrow Donor Association (WMDA) has been accredited by the Dutch Accreditation Council (RvA), allowing the organization to certify stem cell donor registries and transplant coordination services under internationally recognized requirements.
RvA Updates Prisma Reporting Tool for Assessment Reporting
The Dutch Accreditation Council (RvA) has launched an updated version of its Prisma Reporting Tool, introducing changes to how assessment findings, deviations, and reports are structured and presented.
Dutch Healthcare Information Security Standard Updated to Align With ISO 27799
The Royal Netherlands Standardization Institute (NEN) has published an amendment to NEN 7510-2 to align it with ISO 27799,
Netherlands Calls for Stakeholders to Join ISO Tourism Standards Committee
The Netherlands Standardization Institute (NEN) is calling on national stakeholders to take part in international tourism standardization through ISO/TC 228,
Free Risk Tool Launched for Healthcare Information Security Standard NEN 7510
The Netherlands Standardization Institute (NEN) has launched a free risk analysis tool to help healthcare organizations and other entities that process personal health data carry out the information security risk assessment required under NEN 7510.
RvA Publishes INF002 Guidance On ISO 17025 Interpretation
The Dutch Accreditation Council (RvA) has published a new informative document, INF002, explaining how it interprets and applies EN ISO/IEC 17025,
RvA Undergoes EA Peer Evaluation To Confirm MLA Status Under ISO 17011
The Dutch Accreditation Council (RvA) has completed iits 2025 peer evaluation by the European co-operation for Accreditation (EA), a review that determines whether it can remain a signatory to the EA Multilateral Agreement.
NEN Seeks Experts for Clinical Research Standards Committee
The Netherlands Standardization Institute (NEN) is inviting experts to join its Clinical Research standards committee to help shape international standards for clinical investigations of medical devices.
NEN Nearing Full Shift To Digital Standards In February 2026
From February 1, 2026, NEN, the Netherlands’ national standards body, will supply its standards only in digital form, ending the sale of printed standards.
Dutch Stakeholders Invited to Review Water Sampling Standard NEN-EN-ISO 19458
The Netherlands Standardization Institute (NEN) has opened a consultation asking Dutch stakeholders whether the water sampling standard NEN-EN-ISO 19458 should be revised, with input open until February 19, 2026.Global News

ISO 9000 and ISO 19011 Revised: New Guidance for Quality and Auditing
Two cornerstone standards for quality management and auditing have been revised, with the International Organization for Standardization (ISO) publishing new editions of ISO 9000 and ISO 19011.
ISO 9002 Revision Reaches Public Comment Stage
The International Organization for Standardization (ISO) has advanced the revision of ISO 9002 to the Draft International Standard (DIS) stage, opening the project for wider review and feedback.
Global ACI Sets 36-Month Transition for ISO/IEC 17024:2026
The Global Accreditation Cooperation Incorporated (Global ACI) has established a 36-month transition period for ISO/IEC 17024:2026, the newly revised international standard for bodies that certify individuals.
PAS 96:2026 Updates Food Defence Guidance for Emerging Threats
The British Standards Institution (BSI) has published PAS 96:2026, a fully revised edition of its food defence guide.
BRCGS Updates Meat Supply Chain Assurance Module
The British Retail Consortium Global Standards (BRCGS) has updated its Additional Module 11: Meat Supply Chain Assurance.
ISO 29997 Offers First International Framework for Quality Internships
ISO 29997:2025 provides the first international framework designed to help organizations deliver higher-quality internships through clearer planning, management and evaluation practices.
Experts Behind ISO 14001:2026 Explain New Focus on Climate, Biodiversity and Results
The International Organization for Standardization (ISO) has outlined the most important changes in ISO 14001:2026,
New ISO/IEC 15408 and 18045 Editions Update Common Criteria Framework
ISO/IEC 15408-1:2026 and ISO/IEC 18045:2026 have been published, updating the Common Criteria framework, an international system used to evaluate the security of IT products.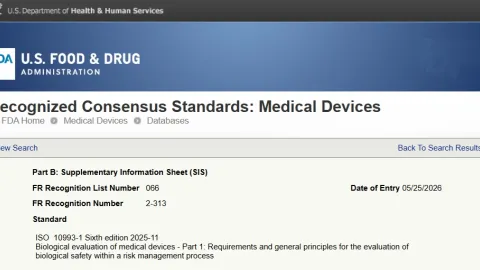
FDA Partially Recognizes ISO 10993-1:2025 for Medical Devices
The U.S. Food and Drug Administration (FDA) has partially recognized ISO 10993-1:2025, the latest edition of the international standard used to assess the biological safety of medical devices.
Licensing Instead of Sales: ISO Changes How Standards Are Used
The International Organization for Standardization (ISO) is preparing to replace its traditional standards sales approach with a licensing model that would give users defined rights to use standards content instead of broad unrestricted access.
BRCGS Updates Food Safety, Packaging, Storage, and Broker Rules to Meet New GFSI Requirements
BRCGS has updated certification requirements for its Food Safety, Packaging Materials, Storage and Distribution, and Agents and Brokers standards,

