News European Union
Please note that you have to be a registered member with paid membership in order to see full articles.
RegisterSelected News
DGQ Releases AI Prompt Library for Quality Managers
The German Society for Quality (DGQ) has released an AI prompt library designed to help quality professionals use artificial intelligence in their daily work.
EU Agrees Common NIS2 Incident Reporting Templates Across Member States
The European Commission (EC), together with EU Member States and the European Union Agency for Cybersecurity (ENISA) through the NIS2 Cooperation Group, has agreed on common templates for reporting cybersecurity incidents.
European AI Logging Standard Opens for Public Comment
CEN and CENELEC have opened the draft standard prEN 18229-1 for public comment, proposing rules for how AI systems should keep records of their activities and decisions under the EU AI Act.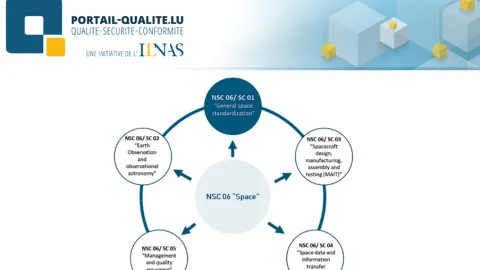
Luxembourg Launches National Space Standardization Commission
The Luxembourg Institute for Standardization, Accreditation, Safety and Quality of Products and Services (ILNAS) has launched a new National Standardization Commission for the space sector,
UNE Marks 40 Years of Standardization With Milestone Video
The Spanish Association for Standardization (UNE) has released a video marking its 40th anniversary,
Sweden Invites Comments on ISO 37401 Diversity Management Standard
The Swedish Institute for Standards (SIS) is inviting stakeholders to comment on ISO/DIS 37401, the draft version of a proposed IISO standard for diversity management.
Industry Coalition Calls for Concrete Steps in DAkkS Reform
The German Federal Ministry for Economic Affairs and Energy (BMWE) has outlined a reform agenda for the German Accreditation Body (DAkkS),
'The Nordic Region Sets the Standard' Initiative Targets Green and Digital Transition
The national standards bodies of Denmark, Finland, Iceland, Norway and Sweden have launched a joint initiative called The Nordic Region Sets the Standard to strengthen cooperation on the green and digital transition.
UNE Presents 2026 Awards for Standardization and Innovation
The Spanish Association for Standardization (UNE) has announced the winners of its 2026 awards, recognizing contributions to innovation, technical standardization and committee leadership.
PCA Marks 25 Years at Warsaw Conference on Accreditation Challenges and Growth
The Polish Centre for Accreditation (PCA) marked its 25th anniversary with a conference in Warsaw that highlighted the role of accreditation in supporting trade, safety and economic development while examining future challenges facing the sector.
Accredia Letter Spotlights Biobanks, ISO 14001 Transition and Procurement
The Italian Accreditation Body (Accredia) has published the May 2026 issue of Accredia Letter,
Environmental Management and Digital Transformation Headline New HZN e-Glasilo
The Croatian Standards Institute (HZN) has published issue 5/2026 of HZN e-Glasilo, with environmental management and digital transformation emerging as the publication's leading themes.Global News

ISO 9000 and ISO 19011 Revised: New Guidance for Quality and Auditing
Two cornerstone standards for quality management and auditing have been revised, with the International Organization for Standardization (ISO) publishing new editions of ISO 9000 and ISO 19011.
ISO 9002 Revision Reaches Public Comment Stage
The International Organization for Standardization (ISO) has advanced the revision of ISO 9002 to the Draft International Standard (DIS) stage, opening the project for wider review and feedback.
Global ACI Sets 36-Month Transition for ISO/IEC 17024:2026
The Global Accreditation Cooperation Incorporated (Global ACI) has established a 36-month transition period for ISO/IEC 17024:2026, the newly revised international standard for bodies that certify individuals.
PAS 96:2026 Updates Food Defence Guidance for Emerging Threats
The British Standards Institution (BSI) has published PAS 96:2026, a fully revised edition of its food defence guide.
BRCGS Updates Meat Supply Chain Assurance Module
The British Retail Consortium Global Standards (BRCGS) has updated its Additional Module 11: Meat Supply Chain Assurance.
ISO 29997 Offers First International Framework for Quality Internships
ISO 29997:2025 provides the first international framework designed to help organizations deliver higher-quality internships through clearer planning, management and evaluation practices.
Experts Behind ISO 14001:2026 Explain New Focus on Climate, Biodiversity and Results
The International Organization for Standardization (ISO) has outlined the most important changes in ISO 14001:2026,
New ISO/IEC 15408 and 18045 Editions Update Common Criteria Framework
ISO/IEC 15408-1:2026 and ISO/IEC 18045:2026 have been published, updating the Common Criteria framework, an international system used to evaluate the security of IT products.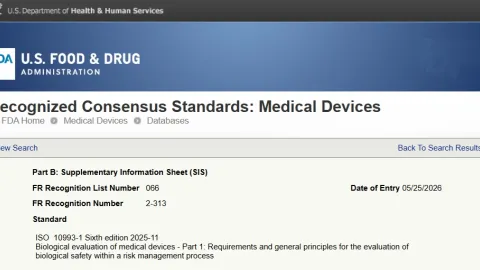
FDA Partially Recognizes ISO 10993-1:2025 for Medical Devices
The U.S. Food and Drug Administration (FDA) has partially recognized ISO 10993-1:2025, the latest edition of the international standard used to assess the biological safety of medical devices.
Licensing Instead of Sales: ISO Changes How Standards Are Used
The International Organization for Standardization (ISO) is preparing to replace its traditional standards sales approach with a licensing model that would give users defined rights to use standards content instead of broad unrestricted access.
BRCGS Updates Food Safety, Packaging, Storage, and Broker Rules to Meet New GFSI Requirements
BRCGS has updated certification requirements for its Food Safety, Packaging Materials, Storage and Distribution, and Agents and Brokers standards,

