News Denmark
Please note that you have to be a registered member with paid membership in order to see full articles.
RegisterSelected News

'The Nordic Region Sets the Standard' Initiative Targets Green and Digital Transition
The national standards bodies of Denmark, Finland, Iceland, Norway and Sweden have launched a joint initiative called The Nordic Region Sets the Standard to strengthen cooperation on the green and digital transition.
Large Danish Companies Face ISO 50001 Certification Deadline
The Danish Standards (DS) is warning companies to prepare for new legal energy management requirements that take effect in Denmark by August 1.
DANAK Updates Customer Portal With Improved Usability and Gradual Enhancements
The Danish Accreditation Fund (DANAK) has introduced a new version of its customer portal,
Danish Standards Celebrate A Century Of Standardization
The Danish national standards body DS is celebrating its 100th anniversary, marking a century of work supporting safety, trade, and innovation in Denmark.
DANAK Updates AMC 15 Guidance On Accreditation Field Visits Including EMAS
The Danish Accreditation Fund (DANAK) has published a revised edition of accreditation message AMC 15, which sets out the scope of DANAK’s field visits used to assess accredited certification bodies and verifiers.
Danish Standards Launches Guide To Prepare Companies For Digital Product Passport
Danish Standards (DS) has launched a new guide to help companies prepare for upcoming EU requirements related to the Digital Product Passport (DPP).
Revised AMC 40 Removes Earlier Restrictions on Accreditation Scopes
The Danish Accreditation Fund, DANAK, has issued a new edition of AMC 40 that removes earlier restrictions that limited which activities certification bodies could cover under their accreditation for quality, environmental, OH&S, and energy management systems.
Denmark Aligns Accreditation Rules for Referral Laboratories and Specialists With ISO 15189
Denmark has revised its national accreditation rules for the use of referral laboratories and external specialists to align with the Danish version of ISO 15189:2022 for medical laboratories.
Danish Standards Introduces AI Search Tool for Standards
Danish Standards (DS) has developed a new AI-based search tool to make standards search easier and more relevant for organizations.
How Denmark Became The Only Country To Certify Its EU Presidency To ISO 20121
Denmark’s EU Council Presidency in the second half of 2025 was certified to ISO 20121, the international standard for sustainable management of events and meetings.
DANAK Passes European Peer Review for Ten Accreditation Areas
DANAK, Denmark’s national accreditation body, has passed its latest peer review by the European co-operation for Accreditation (EA) and remains a full signatory to the European accreditation system.
Danish Version of European Standard EN 45560 Brings Circularity Into Product Design
Denmark has published DS/EN 45560, its national version of the European circular product design standard, giving companies a practical way to integrate circularity into product development from the start.Global News

ISO 9000 and ISO 19011 Revised: New Guidance for Quality and Auditing
Two cornerstone standards for quality management and auditing have been revised, with the International Organization for Standardization (ISO) publishing new editions of ISO 9000 and ISO 19011.
ISO 9002 Revision Reaches Public Comment Stage
The International Organization for Standardization (ISO) has advanced the revision of ISO 9002 to the Draft International Standard (DIS) stage, opening the project for wider review and feedback.
Global ACI Sets 36-Month Transition for ISO/IEC 17024:2026
The Global Accreditation Cooperation Incorporated (Global ACI) has established a 36-month transition period for ISO/IEC 17024:2026, the newly revised international standard for bodies that certify individuals.
PAS 96:2026 Updates Food Defence Guidance for Emerging Threats
The British Standards Institution (BSI) has published PAS 96:2026, a fully revised edition of its food defence guide.
BRCGS Updates Meat Supply Chain Assurance Module
The British Retail Consortium Global Standards (BRCGS) has updated its Additional Module 11: Meat Supply Chain Assurance.
ISO 29997 Offers First International Framework for Quality Internships
ISO 29997:2025 provides the first international framework designed to help organizations deliver higher-quality internships through clearer planning, management and evaluation practices.
Experts Behind ISO 14001:2026 Explain New Focus on Climate, Biodiversity and Results
The International Organization for Standardization (ISO) has outlined the most important changes in ISO 14001:2026,
New ISO/IEC 15408 and 18045 Editions Update Common Criteria Framework
ISO/IEC 15408-1:2026 and ISO/IEC 18045:2026 have been published, updating the Common Criteria framework, an international system used to evaluate the security of IT products.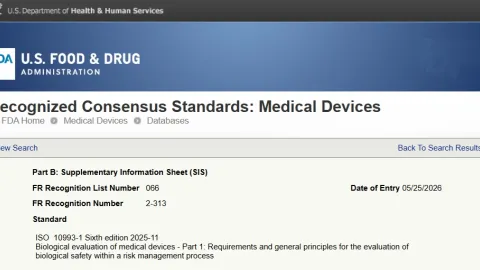
FDA Partially Recognizes ISO 10993-1:2025 for Medical Devices
The U.S. Food and Drug Administration (FDA) has partially recognized ISO 10993-1:2025, the latest edition of the international standard used to assess the biological safety of medical devices.
Licensing Instead of Sales: ISO Changes How Standards Are Used
The International Organization for Standardization (ISO) is preparing to replace its traditional standards sales approach with a licensing model that would give users defined rights to use standards content instead of broad unrestricted access.
BRCGS Updates Food Safety, Packaging, Storage, and Broker Rules to Meet New GFSI Requirements
BRCGS has updated certification requirements for its Food Safety, Packaging Materials, Storage and Distribution, and Agents and Brokers standards,

