News
Please note that you have to be a registered member with paid membership in order to see full articles.
RegisterSelected News
DAkkS Remains In International Accreditation System Even Without Direct Global ACI Membership
The German Accreditation Body (DAkkS) remains effectively part of the global accreditation system through the European co-operation for Accreditation (EA),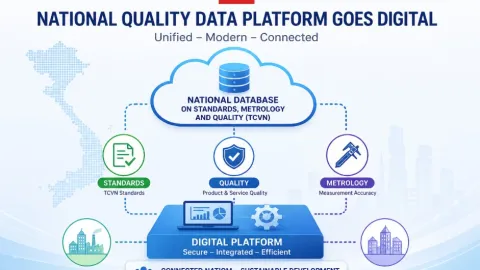
Vietnam Establishes National Database for Standards, Metrology and Quality
The Commission for Standards, Metrology and Quality of Viet Nam (STAMEQ) has defined a unified national database for standards, metrology and quality underDecree 22/2026/ND-CP,
Italy Expands Workplace Safety Prevention Through Free Access To UNI Standards
The Italian Ministry of Labour and Social Policies (MLPS), the National Institute for Insurance against Accidents at Work (INAIL), and the Italian National Standardization Body (UNI) have signed a three-year agreement to provide free access to technical standards on workplace health and safety.
ANSI Starts National Effort To Align Critical Minerals Standards Across The Supply Chain
The American National Standards Institute (ANSI) has launched a multi-phase initiative to coordinate standards for critical minerals, aiming to strengthen U.S. supply chains and reduce reliance on foreign sources.
China Introduces First Commercial Space Standards System Covering Full Industry Chain
The China National Space Administration (CNSA) and the State Administration for Market Regulation (SAMR) have released the Commercial Space Standard System (Version 1.0), China’s first structured framework to guide the country’s growing private space sector.
CyberLink FaceMe Earns ISO 27701 Certification For Privacy Management
CyberLink Corp. has achieved ISO/IEC 27701 certification for its FaceMe facial recognition platform, confirming that its privacy management practices meet international requirements.
China Seeks Experts For International Standards On Battery Recycling And Humanoid Robot Data
The Standards Innovation Department of the State Administration for Market Regulation (SAMR) is inviting experts to join national groups supporting two international standards, one on handling end-of-life power batteries and another on datasets used to train humanoid robots.
Japan Standards Association Opens Applications For 2026 IEC Young Professionals Workshop
The Japanese Standards Association (JSA) has opened applications for the 2026 IEC Young Professionals Workshop, inviting early-career professionals to take part in international standardization activities.
French Quality Day 2026 To Address Skills, Measurement and System Changes
France Qualité, a national association that promotes quality practices in organizations, will hold the 8th French Quality Day on June 9, 2026,
PKN To Host Digital Product Passport Conference In May 2026
The Polish Committee for Standardization (PKN) will host a conference on May 20, 2026 in Warsaw focused on the Digital Product Passport (DPP) and how standards support a digital and sustainable economy.
Quantum Standards Lag Behind Technology Growth, ASCET Scan Finds
The Advancing Standardization for Critical and Emerging Technologies (ASCET) Center of Excellence (CoE) has found in a new environmental scan that quantum standardization is not keeping pace with rapid technological progress,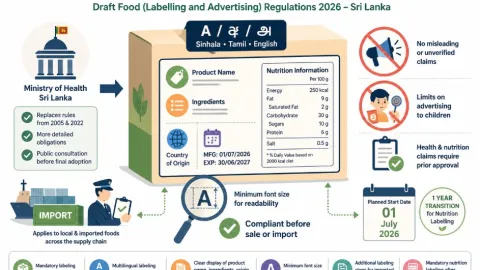
Sri Lanka Proposes Food Labelling And Advertising Rules With Mandatory Nutrition Information
Sri Lanka’s Ministry of Health has issued draft Food (Labelling and Advertising) Regulations 2026 for public consultation.Global News

AutoCarbon Certification Scheme Due in 2027 As Pilot Phase Begins
The International Automotive Oversight Bureau (IAOB) has announced AutoCarbon,
ISO 14064-5 Introduces Guidance For Remote Verification Of Greenhouse Gas Data
The International Organization for Standardization (ISO) has introduced ISO 14064-5, which provides guidance on how to carry out verification and validation of greenhouse gas (GHG) data using remote techniques in digital environments.
ISO 9002 Revision Moves To Draft International Standard Stage
The revision of ISO 9002 has entered the Draft International Standard (DIS) stage, with ISO/TC 176/SC 2, the ISO subcommittee responsible for quality systems, advancing the project into formal consultation.
ISO 37304 Compliance Management Certification Standard Advances to Final Draft Stage
The International Organization for Standardization (ISO) has advanced ISO 37304 to the final draft international standard (FDIS) stage.
One Vote Tips Toward New Edition Of ISO/IEC 17065 Over Amendment
One vote has tipped the decision toward publishing a new edition of ISO/IEC 17065 instead of issuing a separate amendment, following a closely split ballot within the International Organization for Standardization (ISO).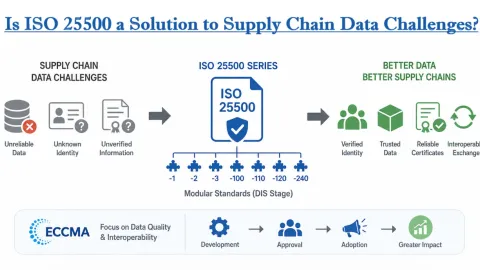
Is ISO 25500 a Solution to Supply Chain Data Challenges?
ISO 25500, a draft series of standards from the International Organization for Standardization (ISO), is presented by the Electronic Commerce Code Management Association (ECCMA) as a possible way to address long-standing problems with unreliable data across global supply chains.
IFS Updates Food, Logistics, Broker And Pacsecure Doctrines To Clarify Certification Rules
The International Featured Standards (IFS) has published updated doctrine documents for its current standards covering food production, logistics, brokerage, and packaging materials, clarifying how certification requirements should be applied without revising the standards themselves.
PAS 25535 on Sustainability in Asset Management Reaches Draft Stage
PAS 25535, a specification focused on integrating sustainability into asset management systems, has reached the committee draft stage within the International Organization for Standardization (ISO), marking the close of its initial comment period.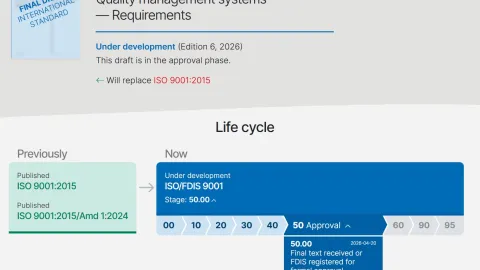
ISO 9001 Revision Moves to FDIS Stage as Early Analysis Questions Scope of Changes
The International Organization for Standardization (ISO) has confirmed that the revision of ISO 9001 has entered the Final Draft International Standard (FDIS) stage, marking the last step before publication.
DIN to Lead ISO IEC Joint Technical Committee 5 on Digital Product Passport Standards
Following an initiative by the German Institute for Standardization (DIN) and the German Commission for Electrical, Electronic & Information Technologies (DKE),
ANSI/TIA-942 Addendum Targets AI Data Center Cooling and High-Density Needs
The Telecommunications Industry Association (TIA) is developing a new addendum to the ANSI/TIA-942 data center standard to address the growing demands of artificial intelligence (AI) infrastructure.

