News Medicine / Pharmaceutical
Please note that you have to be a registered member with paid membership in order to see full articles.
RegisterSelected News

Medsalv Achieves ISO 13485 Certification in New Zealand and Australia
Medsalv, a healthcare company specializing in remanufacturing single-use medical devices, has achieved certification to ISO 13485 across both New Zealand and Australia.
Croatia Completes Transition as All Medical Laboratories Comply With Latest ISO 15189
All 17 accredited medical laboratories in Croatia have successfully demonstrated compliance with HRN EN ISO 15189:2022,
First Medical Biobank Accreditation in Netherlands Underway as RvA Prepares for EA Evaluation
The first medical biobank in the Netherlands has applied for accreditation under ISO 20387.
TÜRKAK Finalizes Transition of Medical Laboratories to Latest ISO 15189 Standard
TÜRKAK has successfully completed the transition of 63 medical laboratories in Turkey to the revised TS EN ISO 15189:2023 standard.
Slavmed Medical Center First in South Caucasus to Earn ISO 7101:2023 Certification
Slavmed Medical Center in Yerevan, Armenia has become the first healthcare provider in the South Caucasus to receive certification for ISO 7101:2023,
Hong Ngoc Hospital Gene Center Accredited to ISO 15189 for Medical Labs
The National Accreditation Bureau (BoA) has accredited the Gene Center at Hong Ngoc General Hospital - Phuc Truong Minh under ISO 15189,
DAkkS Completes ISO 15189 Transition With All Medical Labs in Germany Retaining Accreditation
The Deutsche Akkreditierungsstelle (DAkkS), Germany’s national accreditation body, has successfully completed the nationwide changeover to the revised DIN EN ISO 15189 standard for medical laboratories.
FDA Shift to ISO 13485 Makes Live Quality Management System Essential for Medical Devices
Starting February 2, 2026, the U.S. Food and Drug Administration (FDA) will align its Quality System Regulation with ISO 13485:2016, making it the foundation for all medical device market submissions.
NATA Publishes Biobanking Guide to Strengthen Trust in Stored Biological Materials
The National Association of Testing Authorities (NATA) has released a new Biobanking Industry Guide to help organizations understand the importance of accreditation in maintaining reliable, high-quality biological materials for long-term research and data use.
Cofrac Updates Accreditation Guides for Pathology and Reproductive Biology Labs
The French Accreditation Committee (Cofrac) has released revised versions of two technical accreditation guides, SH GTA 03 and SH GTA 05, aligning them with the 2022 edition of the standard NF EN ISO 15189, the French version of the standard for quality and competence in medical laboratories.
NIST Standard Enables Accurate Dosage of New Anticancer Drugs
The National Institute of Standards and Technology (NIST) has developed the first U.S. standard for measuring the radioactivity of actinium-225, a key ingredient in a new class of anticancer drugs.
Verne Health Earns Triple Certification for Information Security and Quality
Dutch healthcare technology company Verne Health, developer of the Verne EPD Platform for healthcare professionals and hospitals,Global News

PEFC Opens Nominations For Forest Management Working Group Seats
The Programme for the Endorsement of Forest Certification (PEFC) is inviting nominations for its Sustainable Forest Management Working Group, with submissions open until April 22, 2026.
"Global Standard" Opens Second Consultation On Responsible Textile Standard GRTS
Global Standard has opened a second public consultation on Draft 2 of the Global Responsible Textile Standard (GRTS),
DIN SPEC 14027 Establishes First Comprehensive Framework for Corporate Security
The German Institute for Standardization (DIN) has published DIN SPEC 14027,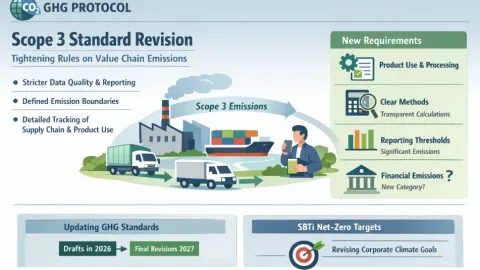
GHG Protocol Begins Scope 3 Revision Tightening Data and Measurement Rules
The Greenhouse Gas Protocol (GHG Protocol) has launched a major revision of its Scope 3 standard,
ISO 17020 Third Edition Published With Risk-Based Approach
A new edition of ISO/IEC 17020 has been published, introducing updated requirements for inspection bodies and replacing the 2012 version after a technical revision.
ISO TS 20125-1 for Eco-Design of Digital Services Released
The International Organization for Standardization (ISO) and the International Electrotechnical Commission (IEC) have published ISO/IEC TS 20125-1:2026,
ISO 19650 Parts 1 and 2 on Information Management in BIM Projects Open for Consultation
The International Organization for Standardization (ISO) has opened a public consultation on proposed changes to ISO 19650 Parts 1 and 2,
TIA Develops First Data Center Infrastructure Quality Standard DCE 9000
The Telecommunications Industry Association (TIA) is developing DCE 9000, the first quality management system (QMS) standard specifically designed for modern data center physical infrastructure, with publication and certification planned for 2027.
ISO 18969 Clinical Evaluation Standard Draft Advances Past Voting Stage
Voting on the Draft International Standard (DIS) for ISO 18969, Clinical evaluation of medical devices, closed on March 10, 2026, marking its progression to stage 40.60 within the International Organization for Standardization (ISO).
GLOBALG.A.P. Replaces Technical Committees With Five Working Groups and Names Members
Agraya has replaced GLOBALG.A.P. technical committees with five technical working groups (TWGs) and appointed members to each group, with up to 16 experts per group across five areas.
Renault Group Updates Customer Requirements for IATF 16949 Suppliers
Renault Group has updated its Customer Specific Requirements (CSR) for IATF 16949:2016, with the new version taking effect in April 2026 and applying to all suppliers across its global operations.

