News France
Please note that you have to be a registered member with paid membership in order to see full articles.
RegisterSelected News

AFNOR Event Brings Together Financial Organizations Holding ‘Committed to CSR’ Label
The French Association for Standardization (AFNOR) brought together financial organizations holding its ‘Committed to CSR’ label during a national meeting in Paris focused on sustainability reporting,
Rhum Bologne First Distillery in Guadeloupe to Earn ISO 22000 Certification
Rhum Bologne, a rum distillery in the French Caribbean territory of Guadeloupe, has received ISO 22000 certification for its food safety management system, becoming the first distillery on the island to achieve the certification.
France Invites Food Sector Feedback on ISO 22000 Revision
The French Association for Standardization (AFNOR) is inviting food sector stakeholders to comment on the draft revision of ISO 22000 as part of both the international update process and the future French adoption of the standard as NF EN ISO 22000.
France Prepares Transition to Revised ISO 17020 Standard
The French Accreditation Committee (Cofrac) is preparing accredited inspection bodies for the transition to the revised ISO/IEC 17020 standard following its publication on March 27, 2026.
French Quality Day 2026 To Address Skills, Measurement and System Changes
France Qualité, a national association that promotes quality practices in organizations, will hold the 8th French Quality Day on June 9, 2026,
Cofrac Updates Map Of Accredited Medical Biology Laboratories In France
The French Accreditation Committee (Cofrac) has released an updated map showing the distribution of accredited medical biology laboratories (LBM) across France as of April 1, 2026.
ISO 22000 Draft Gets French Translation
A French translation of the Draft International Standard (DIS) for ISO 22000, which sets requirements for food safety management systems, has now been completed as part of the revision led by the International Organization for Standardization (ISO).
Burgundy Region Program Helps Firms Prepare for ISO 9001 and ISO 14001 Updates
The Burgundy-Franche-Comté regional branch of the French Quality Movement (MFQ-BFC) has launched a support program to help organizations prepare for upcoming updates to ISO 9001 and ISO 14001.
AFNOR Group Introduces Food Safety Culture Barometer For Food Chain
The AFNOR Group has introduced a dedicated barometer to help organizations in the food chain assess how their teams perceive food safety.
France Qualité Invites Professionals To Contribute To Quality Barometer 2026-2027
France Qualité is inviting quality and performance professionals to take part in the 2026-2027 edition of the Quality Barometer, an international survey designed to track how practices in quality, continuous improvement, and organizational performance are evolving.
AFNOR Report Maps Standards Landscape For Data-Driven Agrifood Systems
AFNOR Group has published a report that maps the emerging standards landscape for data-driven agricultural and agrifood systems, highlighting ongoing international standardization work and the role of French stakeholders in shaping it.
France Qualité Launches Project Group to Develop White Paper on Integrated CSR
France Qualité, a French association that promotes quality management and organizational performance,Global News

ISO 9000 and ISO 19011 Revised: New Guidance for Quality and Auditing
Two cornerstone standards for quality management and auditing have been revised, with the International Organization for Standardization (ISO) publishing new editions of ISO 9000 and ISO 19011.
ISO 9002 Revision Reaches Public Comment Stage
The International Organization for Standardization (ISO) has advanced the revision of ISO 9002 to the Draft International Standard (DIS) stage, opening the project for wider review and feedback.
Global ACI Sets 36-Month Transition for ISO/IEC 17024:2026
The Global Accreditation Cooperation Incorporated (Global ACI) has established a 36-month transition period for ISO/IEC 17024:2026, the newly revised international standard for bodies that certify individuals.
PAS 96:2026 Updates Food Defence Guidance for Emerging Threats
The British Standards Institution (BSI) has published PAS 96:2026, a fully revised edition of its food defence guide.
BRCGS Updates Meat Supply Chain Assurance Module
The British Retail Consortium Global Standards (BRCGS) has updated its Additional Module 11: Meat Supply Chain Assurance.
ISO 29997 Offers First International Framework for Quality Internships
ISO 29997:2025 provides the first international framework designed to help organizations deliver higher-quality internships through clearer planning, management and evaluation practices.
Experts Behind ISO 14001:2026 Explain New Focus on Climate, Biodiversity and Results
The International Organization for Standardization (ISO) has outlined the most important changes in ISO 14001:2026,
New ISO/IEC 15408 and 18045 Editions Update Common Criteria Framework
ISO/IEC 15408-1:2026 and ISO/IEC 18045:2026 have been published, updating the Common Criteria framework, an international system used to evaluate the security of IT products.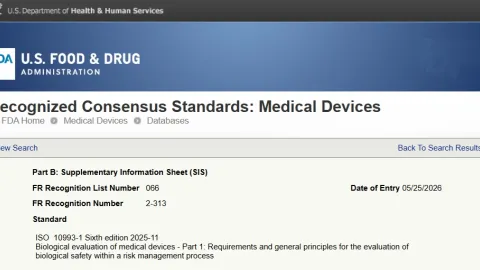
FDA Partially Recognizes ISO 10993-1:2025 for Medical Devices
The U.S. Food and Drug Administration (FDA) has partially recognized ISO 10993-1:2025, the latest edition of the international standard used to assess the biological safety of medical devices.
Licensing Instead of Sales: ISO Changes How Standards Are Used
The International Organization for Standardization (ISO) is preparing to replace its traditional standards sales approach with a licensing model that would give users defined rights to use standards content instead of broad unrestricted access.
BRCGS Updates Food Safety, Packaging, Storage, and Broker Rules to Meet New GFSI Requirements
BRCGS has updated certification requirements for its Food Safety, Packaging Materials, Storage and Distribution, and Agents and Brokers standards,

