News Medicine / Pharmaceutical
Please note that you have to be a registered member with paid membership in order to see full articles.
RegisterSelected News

Consultation Open on Safe Handling of Sterile Medical Equipment in Norway
Standard Norway has opened a consultation on a new technical specification that sets clear requirements for storing, handling, and transporting sterile medical equipment in Norwegian healthcare services.
Russia Now Requires Local Representative for Foreign Medical Device Registration
Foreign manufacturers with medical devices registered in Russia must appoint a Russian Authorized Representative (RUS REP) or update existing representative details by August 31, 2025.
Public Consultation Opens in Norway on Patient Pathway Standard for Diagnostic Imaging
A draft Norwegian version of a new European standard for the patient pathway in diagnostic imaging is now open for public consultation until May 1.
UK to Tighten Post-Market Rules for Medical Devices Starting June 2025
The UK’s new Post-Market Surveillance (PMS) regulations for medical and in vitro diagnostic devices will come into force on June 16, 2025.
NHSGGC Biorepository Becomes First in UK to Earn UKAS Accreditation Under ISO 20387
NHS Greater Glasgow and Clyde's (NHSGGC) Biorepository is the first facility in the UK to receive UKAS accreditation under ISO 20387:2018, the international standard for biobanking.
NEN Seeks Dutch Experts for In Vitro Diagnostics Standards Committee
The Netherlands Standardization Institute (NEN) is seeking Dutch experts to join its In Vitro Diagnostics (IVD) standards committee.
NSAI Seeks Feedback on Potential Revisions to ISO 13485 for Medical Device Quality
The National Standards Authority of Ireland (NSAI) is inviting users of ISO 13485:2016 to share their input on possible revisions through a survey.
Standard Norway Seeks Experts for European Healthcare Standards Group
Standard Norway is inviting 4-6 professionals to join the newly established CEN/CENELEC SAG Healthcare standards group, which aims to identify and address standardization needs in the healthcare sector.
NEN Initiates Development of Sustainability Guidelines for Medical Devices
In April 2025, the Netherlands Standardization Institute (NEN) will begin creating a Dutch Practical Guideline (NPR) focused on the sustainability of medical devices.
Medical Device Industry at Midpoint in Transition to FDA’s ISO 13485-Based QMSR
A year ago, the FDA adopted ISO 13485 into its Quality System Regulation, rebranding it as the Quality Management System Regulation (QMSR).
Topolšica Hospital Achieves Slovenia's First ISO 7101 Certification
Topolšica Hospital has become the first medical institution in Slovenia to earn the ISO 7101 certification for management systems for quality in healthcare organizations.
How EU’s Unique Device Identification Enhances Medical Device Safety
The European Medical Device Regulation (EU) 2017/745 (MDR) requires all medical device manufacturers to assign a Unique Device Identification (UDI) to their products.Global News

ISO Releases Practical Guide To Integrating Multiple Management Systems
ISO has released a new publication, Integrated management systems – A practical guide (IMS PG), offering organizations a clear way to combine multiple management system standards into one coherent system.
ISO TR 25326 Gathers Global Examples Of Green Logistics In Practice
The International Organization for Standardization (ISO) has published ISO/TR 25326:2026, a new international standard that compiles case studies of green logistics to support lower-carbon operations across global supply chains.
BYD Joins IATF As First NEV-Only Automaker And Second Asian Member
The International Automotive Task Force (IATF) has approved the addition of BYD Group as a new member of IATF AISBL,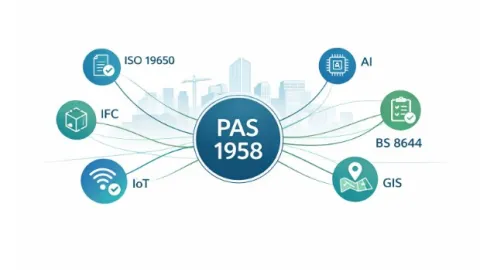
PAS 1958 Published To Connect Built Environment Data Standards
The British Standards Institution (BSI) has published PAS 1958:2026, a new guide that helps organizations understand how existing data and information standards in the built environment, including buildings and infrastructure, work together within a single framework.
International Quality Awards 2026 Finalists Announced
The Chartered Quality Institute (CQI), a professional body for quality management, has announced the finalists for its International Quality Awards 2026,
ASC Chain Of Custody Set To Become Supply Chain Module In Major Overhaul
The Aquaculture Stewardship Council (ASC), an international non-profit that sets standards for responsible seafood farming,
ISO 22000 Revision Enters Public Enquiry Stage
The International Organization for Standardization (ISO) has moved the revision of ISO 22000 to the public enquiry stage.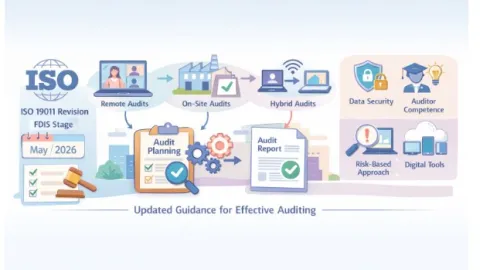
ISO 19011 Auditing Standard Enters Final Stage With Hybrid And Digital Shift
The International Organization for Standardization (ISO) has advanced the revision of ISO 19011 to the Final Draft International Standard (FDIS) stage.
IMDRF Playbook Guides Global Use Of Shared Medical Device Regulatory Reviews
The International Medical Device Regulators Forum (IMDRF) has published a playbook to help regulators use shared approaches when reviewing medical devices, known as regulatory reliance.
DIN Introduces First Framework For AI Testing Tools
The German Institute for Standardization (DIN) has published DIN SPEC 92006, introducing the first unified framework for artificial intelligence (AI) testing tools and setting clear requirements for how these tools should be designed and used.
FSSC 22000 Version 7 Release Moved To May 2026
The Foundation for Food Safety Certification (FSSC) has delayed the launch of FSSC 22000 Version 7, with publication now expected in early May 2026.

