News Australia & Oceania
Please note that you have to be a registered member with paid membership in order to see full articles.
RegisterSelected News
Australia Expands Public Access to Mandatory Standards
The Australian Government has committed Federal Budget support for sponsored public access to mandatory Australian Standards, a move expected to improve access to safety and building requirements for businesses, apprentices, and tradespeople across the country.
Standards Australia Opens Applications for 2027 NEXTgen Program
Standards Australia has opened expressions of interest for its 2027 NEXTgen Program, which gives emerging professionals hands-on experience in standards development through committee participation, training, and industry collaboration.
Australia and Canada Open Applications for IEC Young Professionals Programme 2026
Standards Australia (SA) and the Standards Council of Canada (SCC) have opened applications for the International Electrotechnical Commission (IEC) Young Professionals Programme 2026,
Australia Selects First Teams for International Standards Olympiad
The Standards Australia (SA) has selected two school teams to represent the country at the 21st International Standards Olympiad, marking Australia’s first participation in the event.
Sekuro First Cybersecurity Firm in Australia to Achieve ISO 42001 Certification
Australian cybersecurity firm Sekuro has become the first in Australia and the wider Asia-Pacific region to achieve certification to ISO/IEC 42001, a global standard for managing artificial intelligence.
JASANZ Celebrates 35 Years Of Accreditation Across Australia And New Zealand
The Joint Accreditation System of Australia and New Zealand (JASANZ) is marking 35 years of providing accreditation that helps ensure products, services, and systems meet recognized standards across Australia, New Zealand, and international markets.
JB International Achieves BCI Good Cotton Certification
The Taiwan branch of JB International Co., Ltd. has achieved Better Cotton Initiative (BCI) certification, completing system setup and external verification to align its cotton sourcing with international sustainability requirements.
Quality Business Magazine Reaches Ten-Year Milestone With Anniversary Edition
Quality Business, the quarterly membership magazine of the New Zealand Organisation for Quality (NZOQ), has released a special anniversary edition marking ten years since the publication’s first issue in February 2016.
NATA Opens Revised ISO 17025 Legal And Forensic Appendix For Public Comment
The National Association of Testing Authorities (NATA) has opened public consultation on a revised appendix to its ISO/IEC 17025 application document for legal and forensic science laboratories, with comments due by March 17, 2026.
Australia Set To Enforce New Cybersecurity Rules For Smart Devices
Australia will begin enforcing new mandatory cybersecurity requirements for consumer smart devices on March 4, 2026, as the Cyber Security (Security Standards for Smart Devices) Rules 2025 take effect.
NATA Seeks Experts in Software as Medical Devices and AI for Human Pathology Advisory Committee
The National Association of Testing Authorities, Australia (NATA) is inviting expressions of interest to join its Human Pathology Accreditation Advisory Committee,
Australia Opens Nominations for 2026 Standards Awards
Standards Australia (SA) has opened nominations for the 2026 Standards Awards, marking the start of the next edition of its long-running program that recognizes collaboration and innovation in standards development.Global News

ISO 9001 Revision Moves Into Formal FDIS Ballot Phase
The International Organization for Standardization (ISO) has advanced the revision of ISO 9001 to the next step of the Final Draft International Standard (FDIS) approval process, with the project now listed at stage 50.20 following the earlier 50.00 status.
FSC Opens Consultation on Chain of Custody Revisions as Two Core Standards Reach Final Phase
The Forest Stewardship Council (FSC) has opened consultation on revisions to five Chain of Custody (CoC) documents, with two core standards now entering the final consultation phase.
CanadaGAP Seeks GFSI Recognition for Updated Food Safety Programs
The Global Food Safety Initiative (GFSI) has opened a public stakeholder consultation as CanadaGAP seeks recognition for updated food safety certification programs covering fruit and vegetable operations.
Efforts to Broaden Application of ISO 21378 Underway
Following last year’s confirmation of ISO 21378:2019, a standard for structured audit data collection and exchange, work within ISO/TC 295/WG 1 is now focused on broadening how the standard is applied across organizations.
ISO 14019-3 Working Draft Reviewed Ahead of October Stage Decision
The development of ISO 14019-3, a proposed international standard on sustainability information validation processes,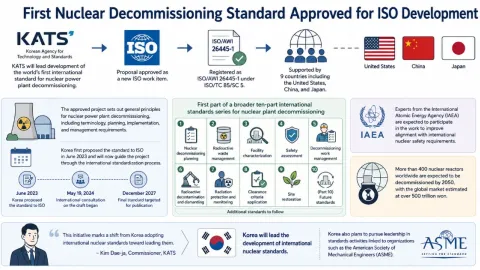
First Nuclear Decommissioning Standard Approved for ISO Development
The Korean Agency for Technology and Standards (KATS) will lead development of the world’s first international standard for nuclear power plant decommissioning after its proposal was approved as a new ISO work item.
Public Consultation Opens for Revised ISO 19650-3 BIM Standard
A public consultation has opened for the revised draft of ISO 19650-3, the part of the ISO 19650 series focused on information management during the operational phase of built assets.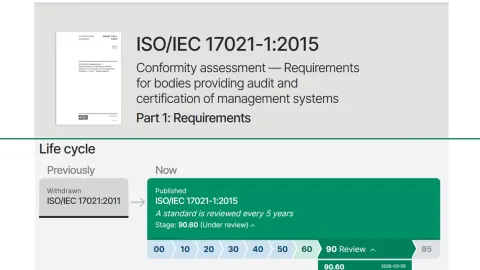
ISO/IEC 17021-1 Confirmation Keeps Certification Rules Stable
The International Organization for Standardization’s (ISO) Committee on Conformity Assessment (ISO/CASCO) has confirmed continuation of the current edition of ISO/IEC 17021-1,
Revised ISO 14024 Aims to Prevent Greenwashing in Ecolabel Programs
The International Organization for Standardization (ISO) has published the third edition of ISO 14024, revising requirements for ecolabel certification programs to improve verification of environmental claims and help prevent greenwashing,
Experts Sought for ISO TS 20008 Food Waste Measurement and Reporting Project
The International Organization for Standardization (ISO) has started development of ISO/AWI TS 20008,
Joint ISO and IEC Team Releases Information Model for SMART Content
The International Organization for Standardization (ISO) and the International Electrotechnical Commission (IEC) have released the first version of the IEC & ISO Information Model for SMART content,