News European Union
Please note that you have to be a registered member with paid membership in order to see full articles.
RegisterSelected News

UNI’s Obiettivo 9001 Platform Adds Two Articles Explaining Key Points and New Features in ISO 9001 Revision
Two new resources explaining the main updates in ISO/DIS 9001 have been released by the Italian Organization for Standardization (UNI) on its Obiettivo 9001 platform.
QI Digital Forum 2025 in Berlin Highlights Shift Toward Digital Quality Infrastructure
The QI Digital Forum 2025, held on October 8 and 9 in Berlin, focused on how a digital quality infrastructure (QI) can accelerate innovation in Germany.
Vienna Testing Department Earns ISO 37301 Compliance Certification
The City of Vienna’s Department MA 39, Austria’s oldest testing, inspection, and certification body, has been certified for its compliance management system under ISO 37301.
Three Finalists Announced for Sweden’s 2025 Standardization Prize
The Swedish Institute for Standards (SIS) has named DigitalWell Arena, the project Renewable 2030 (Förnybart 2030), and the company MannTek as finalists for the 2025 Standardization Prize.
Applications Open for National Industry Awards Well Done in Spain
The Ministry of Industry and Tourism (MINTUR) has launched the 2025 National Industry Awards “Well Done in Spain” to
Italy Updates White Certificate Rules to Reward Energy Efficiency
Italy has revised its white certificates – the country’s main policy tool for promoting energy efficiency through tradable savings certificates.
France Launches Voluntary Environmental Cost Label for Textile Products
France has introduced a new voluntary system for labeling the environmental cost of clothing, effective October 1, 2025.
Three Decades of Quality: Henkel Maribor Marks ISO 9001 Certification Milestone
Henkel Maribor has celebrated 30 years of ISO 9001 certification, reaffirming its long-term commitment to quality management and continuous improvement.
HZN Publishes New E-Newsletter, Major Conferences and Quantum Cybersecurity Highlighted
The Croatian Standards Institute (HZN) released the September 2025 edition of HZN e-Glasilo,
CEN Launches Development of EN 17483-5 on Health Security Services
The European Committee for Standardization (CEN) has begun work on EN 17483-5, a new standard focusing on health security services for the healthcare sector.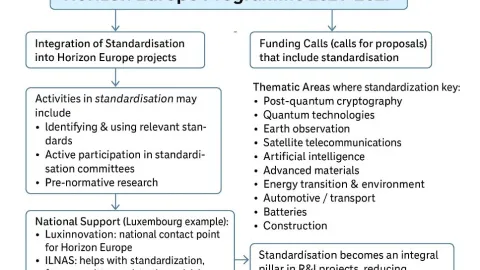
Horizon Europe Calls for Standardization Proposals, Several Closing in November
The European Commission is calling for project proposals that incorporate standardization activities under Horizon Europe, its main program for research and innovation.
European Accreditation Celebrates 25 Years of Strengthening Quality Infrastructure
The European co-operation for Accreditation (EA) is marking its 25th anniversary, celebrating a quarter-century ofGlobal News

IFS Publishes New Doctrine and Product Chart to Support HPC Version 3 Audits
IFS has released two new documents to support audits under the Home and Personal Care (HPC) Version 3 standard:
Small Defense Suppliers Warned to Strengthen Cybersecurity or Risk Losing Contracts
Small and medium enterprises supplying the US Department of Defense are being urged to seek third-party support to meet rising cybersecurity demands under the Cybersecurity Maturity Model Certification (CMMC).
GLOBALG.A.P. Launches IFA v6 Smart Standards for Crops and Propagation Material
GLOBALG.A.P. has released two new product categories under the Integrated Farm Assurance (IFA) version 6 plants scope.
Proposal Aims to Modernize How ISO Standards Are Classified and Organized
Germany’s national standards body, DIN, has proposed the development of ISO/NP TS 26019, which would update the International Classification for Standards (ICS).
Geely’s New Customer Requirements and Scorecard Now in Effect
Geely Holding Group’s updated Customer Specific Requirements (CSRs) and Scorecard Quick Reference Guide officially took effect on July 1, 2025.
Accreditation Opens for Interprivacy Data Protection Scheme Outside EU
Italy’s national accreditation body, Accredia, has opened accreditation for the Interprivacy data protection certification scheme, which applies to the processing of personal data outside the European Union.
IAF Report Reveals 2024 Growth in MLA Coverage and Accreditation Reach
The International Accreditation Forum (IAF) has released its 2024 report on the Multilateral Recognition Arrangement (MLA),
BIFMA Releases 2024 LEVEL e3 Standard Adding Fourth Certification Level and Fixed Criteria
The Business and Institutional Furniture Manufacturers Association (BIFMA) released the 2024 edition of the LEVEL e3 standard on April 21, 2025, starting a 12-month transition period.
ISO 9001 Revision Moves to DIS Stage, Publication Expected by September
The draft of the sixth edition of ISO 9001 is now ready to enter the Draft International Standard (DIS) stage, with publication expected by late August or early September.
ISO/DIS 14001 Released, Refining EMS Scope, Risk Planning and Management Oversight
The Draft International Standard ISO/DIS 14001 has been released, marking a major step in the update of the world’s leading environmental management system (EMS) standard.
ISO 13485 Under Review as Industry Prepares for Potential Revision
The international standard for medical device quality systems, ISO 13485:2016, has entered its mandatory five-year systematic review, which concluded on June 5, 2025.

