News Moldova, Republic of
Please note that you have to be a registered member with paid membership in order to see full articles.
RegisterSelected News

Moldova Moves to Recognize CE-Marked Products in Push for EU Alignment
Moldova is taking a major step toward European Union integration by allowing products with CE marking to enter its market without extra checks.
Serbia Hosts Event to Strengthen Digital Competence of Regional Accreditation Bodies
Serbia’s Chamber of Commerce recently hosted a two-day event aimed to boster the digital capabilities of regional accreditation bodies, focusing on adapting to new technologies.Global News

ISO 9000 and ISO 19011 Revised: New Guidance for Quality and Auditing
Two cornerstone standards for quality management and auditing have been revised, with the International Organization for Standardization (ISO) publishing new editions of ISO 9000 and ISO 19011.
ISO 9002 Revision Reaches Public Comment Stage
The International Organization for Standardization (ISO) has advanced the revision of ISO 9002 to the Draft International Standard (DIS) stage, opening the project for wider review and feedback.
Global ACI Sets 36-Month Transition for ISO/IEC 17024:2026
The Global Accreditation Cooperation Incorporated (Global ACI) has established a 36-month transition period for ISO/IEC 17024:2026, the newly revised international standard for bodies that certify individuals.
PAS 96:2026 Updates Food Defence Guidance for Emerging Threats
The British Standards Institution (BSI) has published PAS 96:2026, a fully revised edition of its food defence guide.
BRCGS Updates Meat Supply Chain Assurance Module
The British Retail Consortium Global Standards (BRCGS) has updated its Additional Module 11: Meat Supply Chain Assurance.
ISO 29997 Offers First International Framework for Quality Internships
ISO 29997:2025 provides the first international framework designed to help organizations deliver higher-quality internships through clearer planning, management and evaluation practices.
Experts Behind ISO 14001:2026 Explain New Focus on Climate, Biodiversity and Results
The International Organization for Standardization (ISO) has outlined the most important changes in ISO 14001:2026,
New ISO/IEC 15408 and 18045 Editions Update Common Criteria Framework
ISO/IEC 15408-1:2026 and ISO/IEC 18045:2026 have been published, updating the Common Criteria framework, an international system used to evaluate the security of IT products.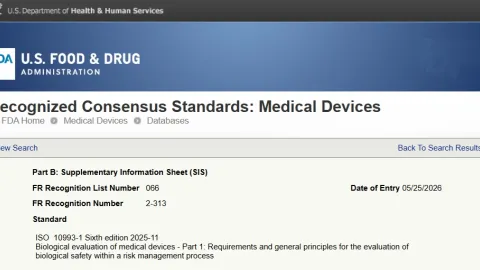
FDA Partially Recognizes ISO 10993-1:2025 for Medical Devices
The U.S. Food and Drug Administration (FDA) has partially recognized ISO 10993-1:2025, the latest edition of the international standard used to assess the biological safety of medical devices.
Licensing Instead of Sales: ISO Changes How Standards Are Used
The International Organization for Standardization (ISO) is preparing to replace its traditional standards sales approach with a licensing model that would give users defined rights to use standards content instead of broad unrestricted access.
BRCGS Updates Food Safety, Packaging, Storage, and Broker Rules to Meet New GFSI Requirements
BRCGS has updated certification requirements for its Food Safety, Packaging Materials, Storage and Distribution, and Agents and Brokers standards,

