News Serbia
Please note that you have to be a registered member with paid membership in order to see full articles.
RegisterSelected News

Serbian Editions of Revised ISO 17020 and ISO 17024 Released
The Institute for Standardization of Serbia (ISS) has published Serbian-language editions of ISO/IEC 17020 and ISO/IEC 17024 following the recent release of the new third editions of both conformity assessment standards.
Sorsix Becomes First in Europe’s Public Health Sector to Earn ISO 42001 Certification
Sorsix has become the first organization in Europe to have governance for a live national healthcare system certified under ISO/IEC 42001, the international standard for AI management systems.
Regional Standards Bodies Discuss SMART Standards and Eurocodes in Sarajevo
The Institute for Standardization of Bosnia and Herzegovina (ISBIH) hosted a regional meeting of national standardization bodies on May 11 and 12 in Sarajevo,
Belgrade to Host Quality and Supply Chain Conference on May 27-28 in organization of Nelt Group
Nelt Group in support of the Belgrade Chamber of Commerce and Industry (PK Belgrade) will hold a two-day conference on quality management and supply chain practices on May 27-28 in Belgrade.
Serbia Opens Public Review of Serbian Translation of Updated ISO 14001
The Institute for Standardization of Serbia (ISS) has opened a public consultation on the draft naSRPS EN ISO 14001:2026, inviting experts to comment on the Serbian translation of the updated environmental management standard.
Serbia Updates Accreditation Requirements for Organic Product Certification Bodies
The Accreditation Body of Serbia (ATS) has announced updates to accreditation scopes for certification bodies in organic food production following the adoption of a new Law on Organic Production, which takes effect on June 1, 2026.
ICT, Telecommunications And Education in New ISS Standardization Bulletin
The Institute for Standardization of Serbia (ISS) has released a new issue of its Standardization bulletin, highlighting how standards support digital infrastructure, education, and management practices.
Serbia Hosts 35th Quality Week Spotlighting Green Transition AI and Risk Management
The 35th Quality Week in Serbia brought together institutions, experts, and industry representatives to explore topics such as green transition, artificial intelligence in healthcare, and risk management in modern organizations.
Academic Standards Day Connects Students With Standards And Career Opportunities In Belgrade
The Institute for Standardization of Serbia (ISS) has announced that Academic Standards Day will take place in Belgrade on April 1,
ISO 21500 On Project Program And Portfolio Management Released In Serbian
Serbia has released SRPS ISO 21500, the national edition of ISO 21500:2021, making guidance on project, program and portfolio management available in Serbian.
Applications Open for Quality Oscar 2026 Business Excellence Awards
Applications are now open for the Quality Oscar (Oskar kvaliteta) 2026, Serbia’s national award for business excellence, recognizing profit and nonprofit organizations based on demonstrated organizational performance and results.
Serbia Adopts Translation of ISO 21001:2025 for Educational Organizations
The KS CASCO Commission has adopted the Serbian translation of ISO 21001:2025, the management system standard for educational organizations.Global News

ISO 9000 and ISO 19011 Revised: New Guidance for Quality and Auditing
Two cornerstone standards for quality management and auditing have been revised, with the International Organization for Standardization (ISO) publishing new editions of ISO 9000 and ISO 19011.
ISO 9002 Revision Reaches Public Comment Stage
The International Organization for Standardization (ISO) has advanced the revision of ISO 9002 to the Draft International Standard (DIS) stage, opening the project for wider review and feedback.
Global ACI Sets 36-Month Transition for ISO/IEC 17024:2026
The Global Accreditation Cooperation Incorporated (Global ACI) has established a 36-month transition period for ISO/IEC 17024:2026, the newly revised international standard for bodies that certify individuals.
PAS 96:2026 Updates Food Defence Guidance for Emerging Threats
The British Standards Institution (BSI) has published PAS 96:2026, a fully revised edition of its food defence guide.
BRCGS Updates Meat Supply Chain Assurance Module
The British Retail Consortium Global Standards (BRCGS) has updated its Additional Module 11: Meat Supply Chain Assurance.
ISO 29997 Offers First International Framework for Quality Internships
ISO 29997:2025 provides the first international framework designed to help organizations deliver higher-quality internships through clearer planning, management and evaluation practices.
Experts Behind ISO 14001:2026 Explain New Focus on Climate, Biodiversity and Results
The International Organization for Standardization (ISO) has outlined the most important changes in ISO 14001:2026,
New ISO/IEC 15408 and 18045 Editions Update Common Criteria Framework
ISO/IEC 15408-1:2026 and ISO/IEC 18045:2026 have been published, updating the Common Criteria framework, an international system used to evaluate the security of IT products.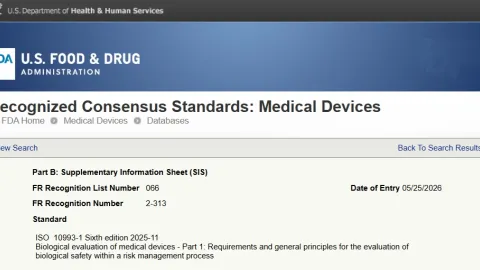
FDA Partially Recognizes ISO 10993-1:2025 for Medical Devices
The U.S. Food and Drug Administration (FDA) has partially recognized ISO 10993-1:2025, the latest edition of the international standard used to assess the biological safety of medical devices.
Licensing Instead of Sales: ISO Changes How Standards Are Used
The International Organization for Standardization (ISO) is preparing to replace its traditional standards sales approach with a licensing model that would give users defined rights to use standards content instead of broad unrestricted access.
BRCGS Updates Food Safety, Packaging, Storage, and Broker Rules to Meet New GFSI Requirements
BRCGS has updated certification requirements for its Food Safety, Packaging Materials, Storage and Distribution, and Agents and Brokers standards,

