News North America
Please note that you have to be a registered member with paid membership in order to see full articles.
RegisterSelected News

Modernizing Accreditation: ANAB Launches Digital Technology Initiative and AI Task Force
The ANSI National Accreditation Board (ANAB) has launched its Digital Technology Initiative (DTI) to advance digital transformation across its accreditation services.
USNC Current Summer Edition Highlights Education in Standards
The new Summer 2025 edition of the USNC Current focuses on how education and training can strengthen the standards workforce and prepare future leaders.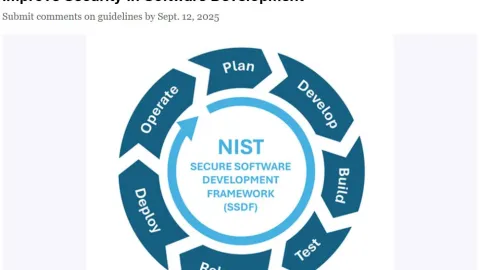
NIST Draft Guidelines Target Stronger Software Security
The U.S. National Institute of Standards and Technology (NIST) has released Special Publication 1800-44, a draft set of practices aimed at improving security throughout the software development process.
Canada Publishes Guides on Accessible Procurement of Goods and Services
Accessibility Standards Canada and Public Services and Procurement Canada have issued two technical guides that provide step-by-step advice on how to integrate accessibility into procurement.
EPA’s WaterSense Program Honored With Ronald H. Brown Standards Leadership Award
The U.S. Environmental Protection Agency (EPA) WaterSense Program will receive the Ronald H. Brown Standards Leadership Award,
New HACCP Model Published to Guide Safe Production of Fermented, Salt-Cured, and Dried Foods
The U.S. Department of Agriculture (USDA) Food Safety and Inspection Service (FSIS) has released a new HACCP model designed to help meat and poultry establishments produce safe ready-to-eat shelf stable fermented, salt-cured, and dried products.
U.S. Standards Strategy 2025 Opens for Public Comment
The American National Standards Institute (ANSI) has released the draft of the 2025 United States Standards Strategy for public review.
National Working Group USA Expands Access to GLOBALG.A.P. Standards
The National Technical Working Group (NTWG) USA has made GLOBALG.A.P. standards more accessible to American producers by tailoring them to local laws and farming practices.
ANSI Seeks Proposals for Interactive Standards Exhibition at Innovation Summit
The American National Standards Institute (ANSI) is calling for proposals to take part in Standards Power Progress, an interactive exhibition at its inaugural Innovation Summit on October 21, 2025, in Rockville, Maryland.
ANSI Names Winners of 2025 Awards for Standards Leadership
The American National Standards Institute (ANSI) has announced the winners of its 2025 Leadership and Service Awards, recognizing 20 individuals for their impact on national and international standardization and certification efforts.
SGS Introduces Services to Support FSMA 204 Food Traceability Compliance
SGS has launched a set of services to help companies prepare for upcoming food traceability requirements in the United States.
FDA Shift to ISO 13485 Makes Live Quality Management System Essential for Medical Devices
Starting February 2, 2026, the U.S. Food and Drug Administration (FDA) will align its Quality System Regulation with ISO 13485:2016, making it the foundation for all medical device market submissions.Global News

International Quality Awards 2026 Finalists Announced
The Chartered Quality Institute (CQI), a professional body for quality management, has announced the finalists for its International Quality Awards 2026,
ASC Chain Of Custody Set To Become Supply Chain Module In Major Overhaul
The Aquaculture Stewardship Council (ASC), an international non-profit that sets standards for responsible seafood farming,
ISO 22000 Revision Enters Public Enquiry Stage
The International Organization for Standardization (ISO) has moved the revision of ISO 22000 to the public enquiry stage.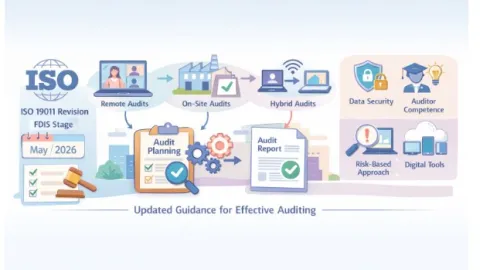
ISO 19011 Auditing Standard Enters Final Stage With Hybrid And Digital Shift
The International Organization for Standardization (ISO) has advanced the revision of ISO 19011 to the Final Draft International Standard (FDIS) stage.
IMDRF Playbook Guides Global Use Of Shared Medical Device Regulatory Reviews
The International Medical Device Regulators Forum (IMDRF) has published a playbook to help regulators use shared approaches when reviewing medical devices, known as regulatory reliance.
DIN Introduces First Framework For AI Testing Tools
The German Institute for Standardization (DIN) has published DIN SPEC 92006, introducing the first unified framework for artificial intelligence (AI) testing tools and setting clear requirements for how these tools should be designed and used.
FSSC 22000 Version 7 Release Moved To May 2026
The Foundation for Food Safety Certification (FSSC) has delayed the launch of FSSC 22000 Version 7, with publication now expected in early May 2026.
FSC Launches Normative Newsfeed For Clearer Requirements Updates
The Forest Stewardship Council (FSC) has introduced a new Normative Newsfeed to make updates to its requirements easier to track and understand.
OECD Report Links Standards To More Effective Regulation
The Organisation for Economic Co-operation and Development (OECD) has released a report examining how governments can improve regulation by making better use of standards, measurement, and assurance systems, known as quality infrastructure (QI).
ISO 9000 Revision Reaches Final Draft Stage, Publication Expected In May 2026
The revision of ISO 9000, the international standard that defines the basic concepts and vocabulary of quality management, has reached the Final Draft International Standard (FDIS) stage, the final step before publication.
ISO and IEC Form JTC 5 Committee on Digital Product Passport Standards
A new international standards committee has been created to develop standards for digital product passports (DPPs), systems designed to share key product information across the entire lifecycle and improve transparency and circular economy practices.

