News North America
Please note that you have to be a registered member with paid membership in order to see full articles.
RegisterSelected News

USDA Campaign Highlights ‘Product of USA’ Label With Full-Origin Requirement
The U.S. Department of Agriculture (USDA) has begun a national campaign to promote its voluntary “Product of USA” label, now limited to meat, poultry, and egg products fully produced in the United States.
Standards Council Of Canada Seeks New Chair To Guide Global Environmental Standards Work
The Standards Council of Canada (SCC) which holds the secretariat for ISO Technical Committee 207 (ISO/TC 207), is recruiting a new Chair for the body responsible for widely used environmental management standards such as the ISO 14000 family.
FDA Schedules Town Hall On Risk Based Inspections Under New QMSR Framework
The U.S. Food and Drug Administration (FDA) has announced a Town Hall on April 1, 2026, to explain how inspections of medical device manufacturers will change under its new Quality Management System Regulation (QMSR), with a focus on a risk-based approach.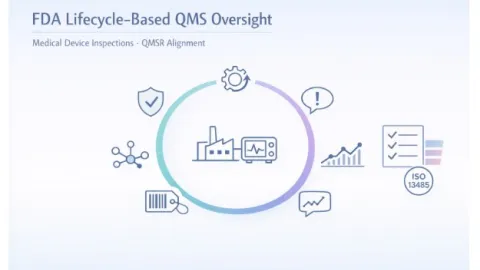
FDA Shifts Medical Device Inspections To Lifecycle-Based QMS Oversight
The U.S. Food and Drug Administration (FDA) has updated how it inspects medical device manufacturers, introducing a lifecycle‑based approach to quality management system oversight under its new Quality Management System Regulation.
ANSI Invites Experts To Lead U.S. TAG For Automation Standards
The American National Standards Institute (ANSI) is inviting experienced automation professionals to consider leadership roles in the U.S. Technical Advisory Group (TAG) to ISO/TC 184/SC 4, which contributes to international standards for automation systems and industrial data.
NIST Launches AI Agent Standards Initiative To Address Security And Interoperability
The National Institute of Standards and Technology (NIST) has launched an AI Agent Standards Initiative to address security, identity, and interoperability challenges as AI agents become more widely used.
GLOBALG.A.P. Tool Aims To Ease Shift From HPSS To IFA Certification
GLOBALG.A.P. has introduced a transition tool to help producers expand their certification from the Harmonized Produce Safety Standard (HPSS) to the Integrated Farm Assurance (IFA) standard, as buyer requirements in North America increasingly extend beyond food safety alone.
FDA Issues New Guidance On Food Traceability Rule
The U.S. Food and Drug Administration (FDA) has released new guidance to help companies comply with its Food Traceability Rule, clarifying how the requirements apply across the food supply chain.
NIST Announces 2025 Baldrige National Quality Award Recipients
Two U.S. health care organizations have been selected to receive the 2025 Malcolm Baldrige National Quality Award, the nation’s only presidential honor for performance excellence.
SQF Institute Names Finalists for 2026 Excellence Awards
The SQF Institute has announced six finalists for its 2026 SQF Excellence Awards, recognizing professionals who advance food safety through auditing and site leadership under the Safe Quality Food program.
ANSI And BSB Introduce ANSI Bharat Compliance Platform For Indian Market
The American National Standards Institute (ANSI) and BSB Edge, a compliance technology company serving regulated industries in India,
Zaire Garcia Wins Terry Burgess Assessor of the Year Award
Zaire Garcia has won the Ninth Annual Terry Burgess Assessor of the Year Award from the ANSI National Accreditation Board (ANAB) for his leadership in assessments and strong commitment to mentoring others.Global News

MSC Opens Review Of Chain Of Custody Standard For Public Consultation
The Marine Stewardship Council (MSC) has launched a public consultation on proposed revisions to its Chain of Custody Standard, inviting stakeholders across the seafood supply chain to provide feedback until May 30, 2026.
Call For Food Sector Stakeholders To Influence ISO 22000 Revision Through National Committees
Food sector stakeholders who apply ISO 22000 in practice have the opportunity to influence its revision by bringing real-world experience into the process.
AutoCarbon Certification Scheme Due in 2027 As Pilot Phase Begins
The International Automotive Oversight Bureau (IAOB) has announced AutoCarbon,
ISO 14064-5 Introduces Guidance For Remote Verification Of Greenhouse Gas Data
The International Organization for Standardization (ISO) has introduced ISO 14064-5, which provides guidance on how to carry out verification and validation of greenhouse gas (GHG) data using remote techniques in digital environments.
ISO 9002 Revision Moves To Draft International Standard Stage
The revision of ISO 9002 has entered the Draft International Standard (DIS) stage, with ISO/TC 176/SC 2, the ISO subcommittee responsible for quality systems, advancing the project into formal consultation.
ISO 37304 Compliance Management Certification Standard Advances to Final Draft Stage
The International Organization for Standardization (ISO) has advanced ISO 37304 to the final draft international standard (FDIS) stage.
One Vote Tips Toward New Edition Of ISO/IEC 17065 Over Amendment
One vote has tipped the decision toward publishing a new edition of ISO/IEC 17065 instead of issuing a separate amendment, following a closely split ballot within the International Organization for Standardization (ISO).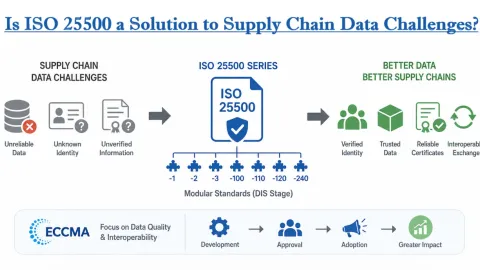
Is ISO 25500 a Solution to Supply Chain Data Challenges?
ISO 25500, a draft series of standards from the International Organization for Standardization (ISO), is presented by the Electronic Commerce Code Management Association (ECCMA) as a possible way to address long-standing problems with unreliable data across global supply chains.
IFS Updates Food, Logistics, Broker And Pacsecure Doctrines To Clarify Certification Rules
The International Featured Standards (IFS) has published updated doctrine documents for its current standards covering food production, logistics, brokerage, and packaging materials, clarifying how certification requirements should be applied without revising the standards themselves.
PAS 25535 on Sustainability in Asset Management Reaches Draft Stage
PAS 25535, a specification focused on integrating sustainability into asset management systems, has reached the committee draft stage within the International Organization for Standardization (ISO), marking the close of its initial comment period.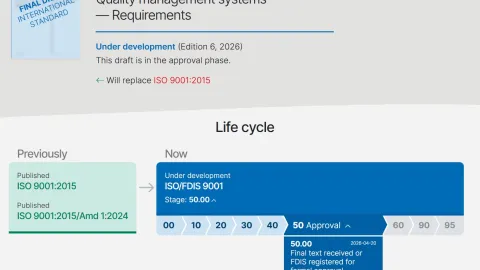
ISO 9001 Revision Moves to FDIS Stage as Early Analysis Questions Scope of Changes
The International Organization for Standardization (ISO) has confirmed that the revision of ISO 9001 has entered the Final Draft International Standard (FDIS) stage, marking the last step before publication.

