News France
Please note that you have to be a registered member with paid membership in order to see full articles.
RegisterSelected News

New CEN-CENELEC Workshop Aims to Improve Repairability in the Automotive Industry
CEN and CENELEC have launched a new workshop to explore how EN 45554 can be applied in the automotive sector, focusing on the repairability of electronic components.
European Report Sets Framework for Sustainable AI Implementation
CEN and CENELEC have published CEN/CLC/TR 18145:2025, a technical report outlining best practices for environmentally sustainable artificial intelligence (AI).
Addressing the Threat of 'Forever Chemicals' Through Standards and Regulations
PFAS, or "forever chemicals" pose a significant environmental and health risk due to their persistence in water, air, and soil.
EU Plans Import Ban on Food Not Meeting Its Standards
The European Commission is set to propose a ban on importing food products that do not comply with EU standards, aiming to protect local farmers.
EFSA Launches EU Model to Assess Pesticide Risks for Honey Bees
The European Food Safety Authority (EFSA) has introduced ApisRAM.03, an advanced model designed to assess the impact of pesticides on honey bee colonies.
Cofrac Announces Transition Plan for PASSI Version 2.2 Cybersecurity Audit Framework
The French Accreditation Committee (Cofrac) has announced the transition plan for version 2.2 of the PASSI (Information Systems Security Audit Service Providers) Requirements Framework, which was officially published by ANSSI on November 29, 2024.
February Certif' Info Issue Features Certification Program Requirements Update
The French Accreditation Committee (Cofrac) has released the February 2025 issue of its "Certif' info" newsletter, highlighting updates to the CERT REF 09 document and focusing on the development and structuring of certification programs.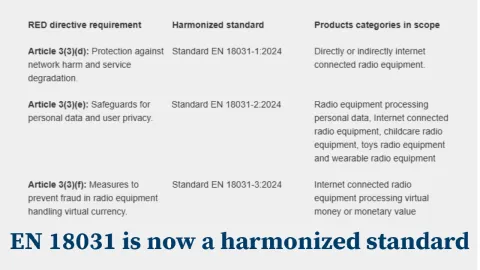
EU Makes EN 18031 a Harmonized Standard Under Radio Equipment Directive
As of January 30, 2025, the European Commission has officially listed the EN 18031 series as a harmonized standard under the Radio Equipment Directive (RED) 2022/2380.
CEN and CENELEC Seek Experts for Industrial Symbiosis Working Groups
CEN and CENELEC, in partnership with DIN, are inviting experts to join Working Groups (WGs) for the development of the Industrial Symbiosis (IS) Standardization Roadmap under the Horizon Europe RISERS project.
CEN and CENELEC Release Brochure on Trusted Standards to Prevent Greenwashing in Environmental Claims
Consumers often struggle to interpret sustainability labels, making it difficult to differentiate genuine environmental efforts from marketing claims.
EU's Competitiveness Compass Underscores Standards’ Role in Global Trade
The European Commission has unveiled the European Union Competitiveness Compass, emphasizing the strategic importance of standards in boosting Europe's role in international trade.
Public Consultation Opens for European Standard on Factory Sustainability
A draft European standard for assessing factory sustainability is now open for public consultation until March 10.Global News

IFS Publishes New Guideline to Support Wholesale and Cash & Carry Audits
IFS has released a new guideline to help companies prepare for audits under the IFS Wholesale and Cash & Carry 3 Standard.
Shift to Green Coding Responds to Rising Energy and Environmental Pressure
Green coding, the practice of designing and writing software that uses less energy, is becoming an important part of modern software development.
IATF Releases New Updates to Rules 6th Edition and IATF 16949
The International Automotive Task Force (IATF) has approved Stakeholder Communiqué SC-2025-003, announcing new Sanctioned Interpretations and Frequently Asked Questions for both the Rules 6th Edition and the automotive quality standard IATF 16949.
Codex Commission’s 48th Session Confirms Updates Across Core Food Safety Standards
The Codex Alimentarius Commission adopted major changes to international food-safety rules during its 48th session in Rome from November 10 to 14.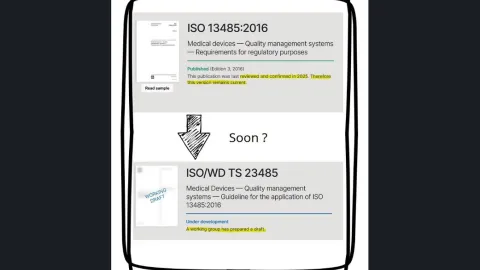
ISO 13485 Application Guide, TS 23485, Under Way After Standard Confirmed as Current
A new guide on how to apply ISO 13485 is moving through development shortly after the standard was reconfirmed in its 2016 version (no revision will be done), despite remaining unaligned with the Annex SL structure.
A Different Approach: China Strengthens Oversight of Certification Bodies from 2026
China has issued new regulations that directly target accredited certification bodies that ignore international requirements, and the new rules will take effect on January 1, 2026.
ISO and IEC Develop Global Standard to Embed Ecodesign in Medical Devices
The International Organization for Standardization (ISO) and the International Electrotechnical Commission (IEC) are developing a new global standard to formally integrate ecodesign into the development of medical devices.
IFS Releases Updated Guideline for Effective Foreign Body Management
International Featured Standards (IFS) has issued an updated Guideline for an Effective Foreign Body Management,
Germany Heads Most ISO Secretariats But China Rapidly Gains Ground
Germany remains the world’s top country in international standardization, holding 17.1% of all ISO secretariats, according to the 2025 International Standardization Barometer published by the German Institute for Standardization (DIN).
Europe Now Conditionally Accepts FDA Inspections Conducted Abroad
The European Medicines Agency (EMA) and the U.S. Food and Drug Administration (FDA) have entered a new stage of cooperation under their Mutual Recognition Agreement (MRA) for Good Manufacturing Practice (GMP) inspections.
New International Standard in Development for Microbial Cultures and Probiotics
Work has begun within ISO to create a global standard that defines how microorganisms used in food and feed, such as bacteria, yeasts, and fungi, should be characterized and quality assured.

