News Netherlands
Please note that you have to be a registered member with paid membership in order to see full articles.
RegisterSelected News

Netherlands Takes the Reins on Global Carbon Capture, Transport, and Storage Standards
The Netherlands is driving international standardization of Carbon Capture, Utilisation and Storage (CCUS), with the Dutch Standardization Institute NEN coordinating efforts through the CEN/TC 474 committee.
New RvA Document Clarifies Rules for Flexible Accreditation Scopes in Netherlands
The Accreditation Council of the Netherlands (RvA) has published a new document, RvA-T054, that explains how flexible scopes of accreditation should be handled.
NEN Seeks Dutch Experts to Explore National Standards for Ayurveda and Yoga
The Netherlands is exploring whether there is enough interest to develop national standards for Ayurveda and yoga.
Zwolle Breaks Ground with First PEFC Trees outside Forests Certification
The municipality of Zwolle in the Netherlands has become the first to receive the Programme for the Endorsement of Forest Certification (PEFC) Trees outside Forests (TOF) certificate, marking a milestone in sustainable tree management beyond traditional forests.
NEN Seeks Dutch Experts for In Vitro Diagnostics Standards Committee
The Netherlands Standardization Institute (NEN) is seeking Dutch experts to join its In Vitro Diagnostics (IVD) standards committee.
NEN Initiates Development of Sustainability Guidelines for Medical Devices
In April 2025, the Netherlands Standardization Institute (NEN) will begin creating a Dutch Practical Guideline (NPR) focused on the sustainability of medical devices.
Dutch Experts Sought for New 'Chain of Custody' Standards
The Netherlands Standardization Institute (NEN) is seeking Dutch experts to contribute to the development of new 'chain of custody' standards, specifically ISO 13662 'Mass Balance' and ISO 13659 'Book and Claim'.
Dutch Involvement in European Sustainability Standards: Strong Foundation, But More Industry Support Essential
In a recent interview, Emiel Janssen, consultant at NEN, discussed the Netherlands’ role in shaping European sustainability standards.
Dutch Experts Sought for New European Circular Construction Standards
The European standards committee CEN/TC 350/SC 1 has established five new working groups to develop standards for circular design, material efficiency, and reuse in the construction sector.
Dutch Energy Sector Pushes for Circular Infrastructure as NEN Supports Transition
The Dutch energy sector is taking significant steps toward a circular economy to reduce the use of raw materials like aluminum, copper, and steel.
Dutch Initiative Aims to Close Standards Gap in Responsible Risk Profiling Algorithms
NEN, the Dutch standardization institute, is launching the development of a Dutch Technical Agreement (NTA) to guide organizations in the responsible use of risk profiling algorithms.
NEN Seeks Public Input on Revised Non-Standard Emission Measurement Guideline NPR 8117
The Netherlands Standardization Institute (NEN) is inviting stakeholders to provide feedback on the proposed revision of NPR 8117,Global News

BYD Joins IATF As First NEV-Only Automaker And Second Asian Member
The International Automotive Task Force (IATF) has approved the addition of BYD Group as a new member of IATF AISBL,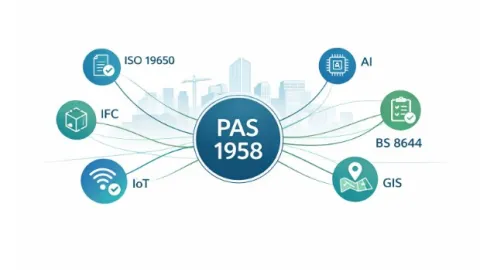
PAS 1958 Published To Connect Built Environment Data Standards
The British Standards Institution (BSI) has published PAS 1958:2026, a new guide that helps organizations understand how existing data and information standards in the built environment, including buildings and infrastructure, work together within a single framework.
International Quality Awards 2026 Finalists Announced
The Chartered Quality Institute (CQI), a professional body for quality management, has announced the finalists for its International Quality Awards 2026,
ASC Chain Of Custody Set To Become Supply Chain Module In Major Overhaul
The Aquaculture Stewardship Council (ASC), an international non-profit that sets standards for responsible seafood farming,
ISO 22000 Revision Enters Public Enquiry Stage
The International Organization for Standardization (ISO) has moved the revision of ISO 22000 to the public enquiry stage.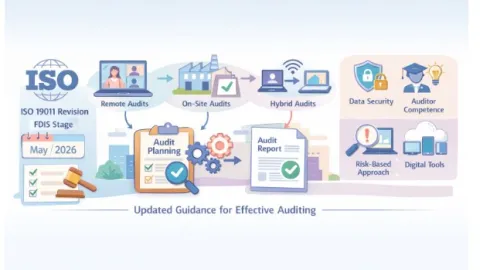
ISO 19011 Auditing Standard Enters Final Stage With Hybrid And Digital Shift
The International Organization for Standardization (ISO) has advanced the revision of ISO 19011 to the Final Draft International Standard (FDIS) stage.
IMDRF Playbook Guides Global Use Of Shared Medical Device Regulatory Reviews
The International Medical Device Regulators Forum (IMDRF) has published a playbook to help regulators use shared approaches when reviewing medical devices, known as regulatory reliance.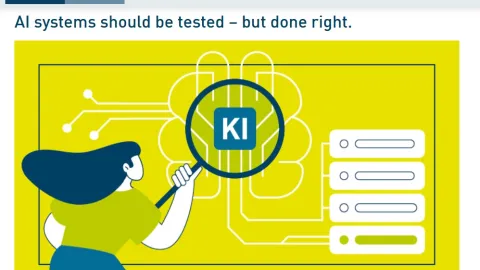
DIN Introduces First Framework For AI Testing Tools
The German Institute for Standardization (DIN) has published DIN SPEC 92006, introducing the first unified framework for artificial intelligence (AI) testing tools and setting clear requirements for how these tools should be designed and used.
FSSC 22000 Version 7 Release Moved To May 2026
The Foundation for Food Safety Certification (FSSC) has delayed the launch of FSSC 22000 Version 7, with publication now expected in early May 2026.
FSC Launches Normative Newsfeed For Clearer Requirements Updates
The Forest Stewardship Council (FSC) has introduced a new Normative Newsfeed to make updates to its requirements easier to track and understand.
OECD Report Links Standards To More Effective Regulation
The Organisation for Economic Co-operation and Development (OECD) has released a report examining how governments can improve regulation by making better use of standards, measurement, and assurance systems, known as quality infrastructure (QI).

