News Namibia
Please note that you have to be a registered member with paid membership in order to see full articles.
RegisterGlobal News

MSC Opens Review Of Chain Of Custody Standard For Public Consultation
The Marine Stewardship Council (MSC) has launched a public consultation on proposed revisions to its Chain of Custody Standard, inviting stakeholders across the seafood supply chain to provide feedback until May 30, 2026.
Call For Food Sector Stakeholders To Influence ISO 22000 Revision Through National Committees
Food sector stakeholders who apply ISO 22000 in practice have the opportunity to influence its revision by bringing real-world experience into the process.
AutoCarbon Certification Scheme Due in 2027 As Pilot Phase Begins
The International Automotive Oversight Bureau (IAOB) has announced AutoCarbon,
ISO 14064-5 Introduces Guidance For Remote Verification Of Greenhouse Gas Data
The International Organization for Standardization (ISO) has introduced ISO 14064-5, which provides guidance on how to carry out verification and validation of greenhouse gas (GHG) data using remote techniques in digital environments.
ISO 9002 Revision Moves To Draft International Standard Stage
The revision of ISO 9002 has entered the Draft International Standard (DIS) stage, with ISO/TC 176/SC 2, the ISO subcommittee responsible for quality systems, advancing the project into formal consultation.
ISO 37304 Compliance Management Certification Standard Advances to Final Draft Stage
The International Organization for Standardization (ISO) has advanced ISO 37304 to the final draft international standard (FDIS) stage.
One Vote Tips Toward New Edition Of ISO/IEC 17065 Over Amendment
One vote has tipped the decision toward publishing a new edition of ISO/IEC 17065 instead of issuing a separate amendment, following a closely split ballot within the International Organization for Standardization (ISO).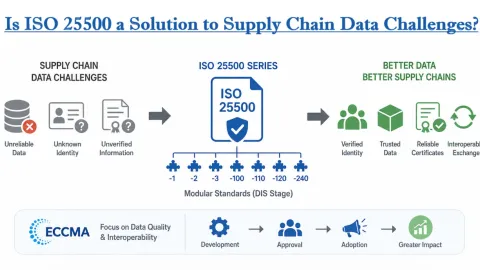
Is ISO 25500 a Solution to Supply Chain Data Challenges?
ISO 25500, a draft series of standards from the International Organization for Standardization (ISO), is presented by the Electronic Commerce Code Management Association (ECCMA) as a possible way to address long-standing problems with unreliable data across global supply chains.
IFS Updates Food, Logistics, Broker And Pacsecure Doctrines To Clarify Certification Rules
The International Featured Standards (IFS) has published updated doctrine documents for its current standards covering food production, logistics, brokerage, and packaging materials, clarifying how certification requirements should be applied without revising the standards themselves.
PAS 25535 on Sustainability in Asset Management Reaches Draft Stage
PAS 25535, a specification focused on integrating sustainability into asset management systems, has reached the committee draft stage within the International Organization for Standardization (ISO), marking the close of its initial comment period.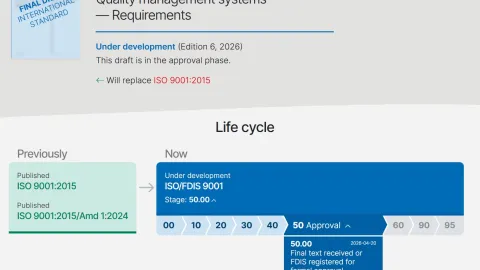
ISO 9001 Revision Moves to FDIS Stage as Early Analysis Questions Scope of Changes
The International Organization for Standardization (ISO) has confirmed that the revision of ISO 9001 has entered the Final Draft International Standard (FDIS) stage, marking the last step before publication.

