News
Please note that you have to be a registered member with paid membership in order to see full articles.
RegisterSelected News

ASQA Signs $46 Million Laboratory Expansion Deal With TCRC
The Afghanistan Standards and Quality Authority (ASQA) has signed a 46 million-dollar deal with Indian company TCRC to expand and equip quality assurance laboratories in Kabul and nine border points across Afghanistan over the next five years.
TÜRKAK Joins International AIQI Consortium
The Turkish Accreditation Agency (TÜRKAK) has joined the AIQI Consortium, an international initiative focused on strengthening quality infrastructure and governance for artificial intelligence (AI).
Australia Expands Public Access to Mandatory Standards
The Australian Government has committed Federal Budget support for sponsored public access to mandatory Australian Standards, a move expected to improve access to safety and building requirements for businesses, apprentices, and tradespeople across the country.
Indonesia Revises Good Cosmetic Manufacturing Practice Certification Rules
Indonesia’s National Agency of Drug and Food Control (BPOM) has issued new rules for Good Cosmetic Manufacturing Practice (CPKB) certification,
CJ CheilJedang First in South Korea to Introduce BRCGS Food Safety Culture Module
CJ CheilJedang, one of South Korea’s largest food companies, has become the first company in South Korea to implement the BRCGS Food Safety Culture Excellence (FSCE) module, using the program to assess food safety culture and identify areas for improvement across the company.
SCC Guide Helps Municipalities Across the Country Apply ISO 14092 Climate Adaptation Standard
The Standards Council of Canada (SCC) has released a new application guide to help municipalities across the country apply ISO 14092:2026 to climate adaptation planning.
Inmetro Opens Pilot Accreditation Program for Brazilian Biobanks
The National Institute of Metrology, Quality and Technology (Inmetro) has opened applications for a pilot accreditation program for biobanks in Brazil based on ISO 20387.
Polish Accreditation Centre Opens CBAM Verification Program
The Polish Accreditation Centre (PCA) has launched a new accreditation process for bodies that verify emissions data under the European Union’s Carbon Border Adjustment Mechanism (CBAM).
South Korea Revises Two Good Recycled Product (GR) Quality Certification Standards
The Korean Agency for Technology and Standards (KATS) has opened a public consultation on proposed revisions to two Good Recycled Product (GR) quality certification standards covering reinforcing steel bars and rubber fire-stop filling materials used in electrical installations.
Students Invited to Explore a World Without Standards in ANSI Paper Competition
The American National Standards Institute (ANSI) is reminding high school and college students across the United States to enter its 2025/2026 Student Paper Competition by June 5, 2026.
BIS Standards-Excellence Awards Presented to IIT Roorkee Students
The Bureau of Indian Standards (BIS) and the Indian Institute of Technology Roorkee (IIT Roorkee) presented the BIS Standards-Excellence Awards on May 13 to students and researchers at IIT Roorkee.
‘Day of ÚNMZ and ČAS’ Conference Highlights Data and Digitalization in Cities and Construction
The Czech Office for Standards, Metrology and Testing (ÚNMZ) and the Czech Standardization Agency (ČAS) held the “Day of ÚNMZ and ČAS” conference in Prague on May 14,Global News

ISO 9001 Revision Moves Into Formal FDIS Ballot Phase
The International Organization for Standardization (ISO) has advanced the revision of ISO 9001 to the next step of the Final Draft International Standard (FDIS) approval process, with the project now listed at stage 50.20 following the earlier 50.00 status.
FSC Opens Consultation on Chain of Custody Revisions as Two Core Standards Reach Final Phase
The Forest Stewardship Council (FSC) has opened consultation on revisions to five Chain of Custody (CoC) documents, with two core standards now entering the final consultation phase.
CanadaGAP Seeks GFSI Recognition for Updated Food Safety Programs
The Global Food Safety Initiative (GFSI) has opened a public stakeholder consultation as CanadaGAP seeks recognition for updated food safety certification programs covering fruit and vegetable operations.
Efforts to Broaden Application of ISO 21378 Underway
Following last year’s confirmation of ISO 21378:2019, a standard for structured audit data collection and exchange, work within ISO/TC 295/WG 1 is now focused on broadening how the standard is applied across organizations.
ISO 14019-3 Working Draft Reviewed Ahead of October Stage Decision
The development of ISO 14019-3, a proposed international standard on sustainability information validation processes,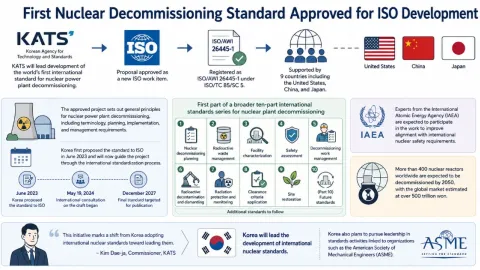
First Nuclear Decommissioning Standard Approved for ISO Development
The Korean Agency for Technology and Standards (KATS) will lead development of the world’s first international standard for nuclear power plant decommissioning after its proposal was approved as a new ISO work item.
Public Consultation Opens for Revised ISO 19650-3 BIM Standard
A public consultation has opened for the revised draft of ISO 19650-3, the part of the ISO 19650 series focused on information management during the operational phase of built assets.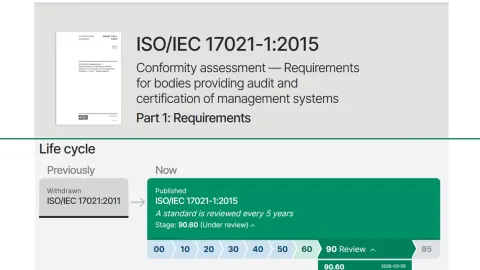
ISO/IEC 17021-1 Confirmation Keeps Certification Rules Stable
The International Organization for Standardization’s (ISO) Committee on Conformity Assessment (ISO/CASCO) has confirmed continuation of the current edition of ISO/IEC 17021-1,
Revised ISO 14024 Aims to Prevent Greenwashing in Ecolabel Programs
The International Organization for Standardization (ISO) has published the third edition of ISO 14024, revising requirements for ecolabel certification programs to improve verification of environmental claims and help prevent greenwashing,
Experts Sought for ISO TS 20008 Food Waste Measurement and Reporting Project
The International Organization for Standardization (ISO) has started development of ISO/AWI TS 20008,
Joint ISO and IEC Team Releases Information Model for SMART Content
The International Organization for Standardization (ISO) and the International Electrotechnical Commission (IEC) have released the first version of the IEC & ISO Information Model for SMART content,