Blogs
Please note that you have to be a registered member with paid membership in order to see full articles.
Register
AI Tools Gain Ground in ISO 9001 Internal Audits
Artificial intelligence (AI) tools are increasingly being integrated into ISO 9001 internal audits to automate planning, analyze larger datasets, improve reporting, and support risk-based auditing aligned with ISO 19011, the international guidance standard for management systems auditing.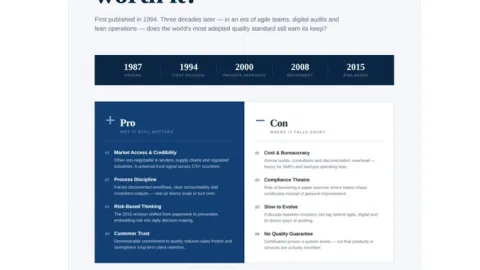
Is ISO 9001 Certification Worth It? It Depends
ISO 9001 still delivers value, but only for companies that use it to improve how they operate rather than treating certification as the final objective.
Diary of a First ISO 42001 Implementation: Challenges of Building an AI Management System
Implementing ISO/IEC 42001:2023 for the first time can involve technical, organizational, and emotional challenges, as shown in a personal account following the effort to build an AI management system inside a regional technology company.
Auditors Do Not Need to Dominate the Room to Be Effective
Maintaining strong standards and effective compliance work does not mean that auditors need to be overly confrontational.
7 Reasons Workers Bypass Management Systems
Employees often ignore management systems not out of resistance, but because the systems are disconnected from real daily work, writes quality coach and mentor Ekaterina Potemkina.
Critical Thinking as a Missing Skill in Audits
Critical and analytical thinking remain underdeveloped skills in many audit environments, writes Jörg Westphal, managing partner at Hellmund Die Personalberater.
Keeping Up With ISO 22000 Changes Through a Tagging System
Food safety professionals are facing growing pressure to keep up with rapid changes in food safety standards and certification schemes amid the ongoing ISO 22000 revision and related food safety updates.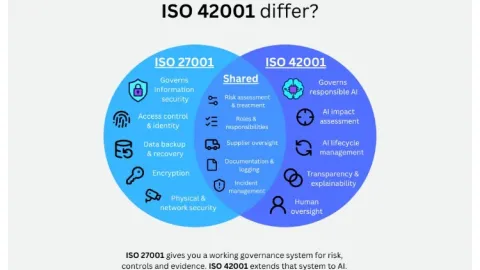
Companies Certified to ISO 27001 May Be Closer to ISO 42001 Than They Think
Organizations certified to ISO 27001 information security management systems (ISMS) may already have much of the governance structure needed for ISO 42001 AI management systems (AIMS) in place.
Understanding Flexible Scope in Accreditation
Who decides whether a conformity assessment body can operate under a flexible scope in accreditation?
How Food Labels Shape Consumer Eating Habits
Food labels are increasingly being linked to healthier consumer choices and lower purchases of ultra-processed foods (UPFs), as governments and researchers explore ways to address rising obesity rates and diet-related health problems.
If Only Auditors Were Stacked on Shelves
If certification bodies worked like grocery stores, with auditors sitting ready on shelves whenever needed, life would certainly be easier for companies.
ISO 19011 Revision May Matter More Than ISO 9001 for Future Audits
ISO 19011:2026 could have a bigger practical impact on companies than the upcoming ISO 9001 revision because it will reshape how organizations prepare for audits and prove their management systems are effective.
How to Prevent Leading Forensic Lab Nonconformities
The ANSI National Accreditation Board (ANAB) identified 548 nonconformities across 543 forensic testing and calibration assessments in 2025,
Preparing for Ethics and Quality Culture Requirements in ISO 9001:2026
New ethics and quality culture requirements introduced in the Final Draft International Standard (FDIS) of ISO 9001 do not explicitly call for organizations to maintain formal evidence, yet
Quality Professionals Must Shape Their Role Before It Shapes Them
Quality professionals can become overwhelmed and lose focus when they do not actively define their role, a point raised by Ekaterina Potemkina of QUALITY-ALL-IN in an opinion piece.
Closing CAPAs Without Effectiveness Checks Fails to Prove Issues Are Fixed
Even a major medical device manufacturer with ISO 13485 certification can run CAPA processes without meaningful effectiveness checks, as highlighted by Michelle Hilling.
ISO 9001 As A Mirror For Exposing Hidden Process Gaps
Companies sometimes pursue ISO 9001 certification to meet customer demands, but even such motivation can lead to unexpected benefits once a quality management system is properly implemented.
Reasons Why Certification Audits Lose Their Edge
Certification audits can either challenge and strengthen organizations or simply help them tick the box, and concern is growing that the latter is becoming more common.
Missed Opportunity To Fix Known Issues: Where ISO 9001:2026 Falls Short
The ongoing revision of ISO 9001 introduces few meaningful changes to requirements and leaves known issues unresolved,
Reaching Quality Maturity Requires Simpler, More Effective Systems
Many quality systems become heavier over time, but maturity comes from making them work better, not adding more layers.
Quality Issues As Symptoms Of Weak Management Systems
What is often described as a quality issue may instead point to a weakness in how the organization is managed.
Audit Theater Wastes Time And Hides Real Problems
Companies often stage their systems before audits instead of showing how they actually work.
Firefighting Signals Missing Alignment Even In ISO Certified Systems
Organizations can remain stuck in constant firefighting despite ISO certification, pointing to a lack of alignment between systems and daily work.

